Click the image below to view the animated version of this
cover.
Created by: Kishore Hari
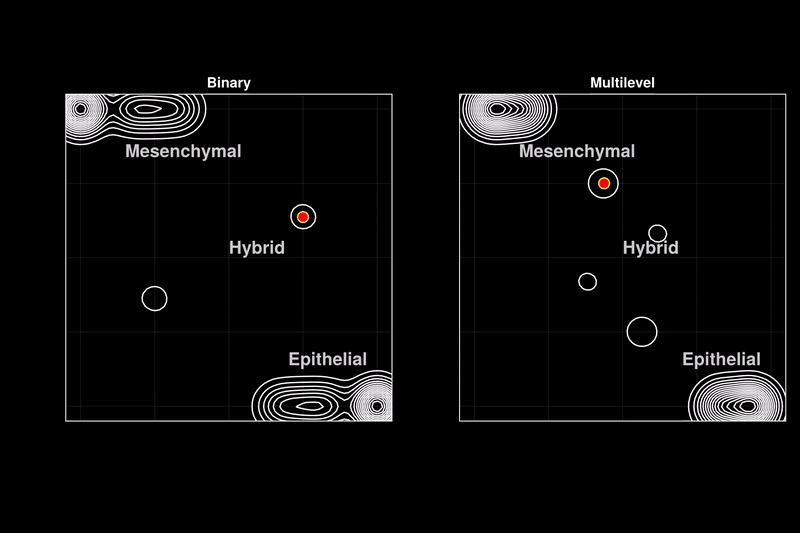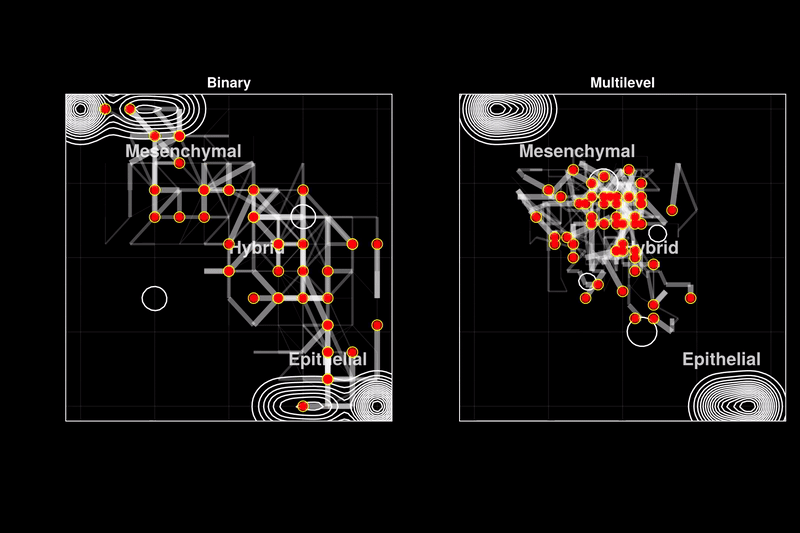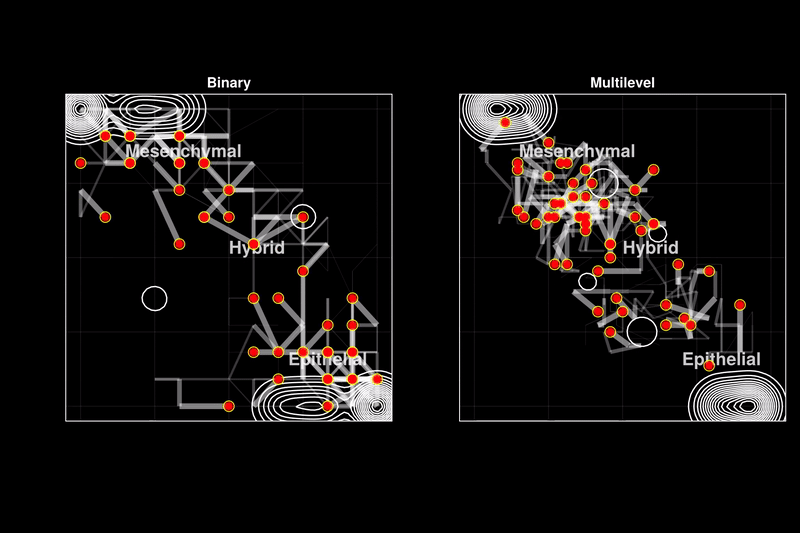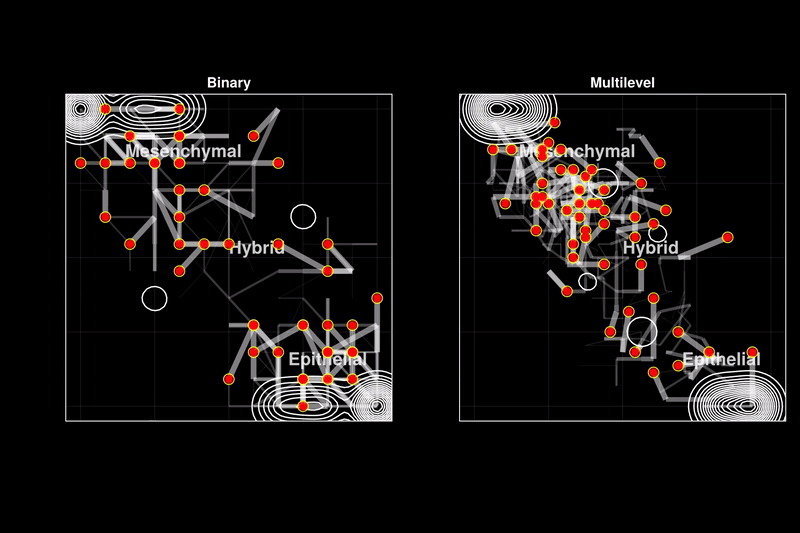
Issue 361: Hybrid epithelial-mesenchymal phenotypes have emerged as crucial components of cancer metastasis, as they enhance many pro-metastatic characteristics of cancer cells, including collective migration, stemness, and therapy resistance. A key characteristic of these hybrid phenotypes is the extensive heterogeneity in gene expression observed in cancer. The drivers, mechanisms, and implications of this heterogeneity for the stability of hybrid states remain highly debated. Using discrete models of gene regulatory networks, we show that stable hybrid states can be achieved through multiple stable expression levels across network genes, as opposed to the binary expression levels permitted in standard Boolean models. Multilevel models reveal that the network structure inherently supports stable hybrid states. While the relative abundance of hybrid phenotypes does not vary significantly in weakly stochastic environments expected in cell cultures or non-pathological conditions (blue ovals), stochastic environments expected in tumors and metastasis lead to a stable 'hybrid-cloud' in the multilevel model, allowing cells to retain the pro-metastatic hybrid phenotype while dynamically shifting their expression patterns (red dots with black trajectories). Thus, we propose stable intermediate expression levels as a mechanism underlying the heterogeneous expression patterns observed in hybrid E/M phenotypes, providing cancer cells with a dynamic survival strategy through metastasis.
