Mathematical models for cancer immunotherapies
Behind the Paper
Mathematical model of a personalized neoantigen cancer vaccine and the human immune system
Marisabel Rodriguez Messan, Osman N. Yogurtcu, Joseph R. McGill, Ujwani Nukala, Zuben E. Sauna, Hong Yang
Read the paperMathematical Modeling of Neoantigen Cancer Vaccines: A Multifaceted Approach
Neoantigen cancer vaccination shows promise as a cancer treatment by stimulating T cells to selectively eliminate cancer cells. However, the underlying processes and mechanisms are complex. In order to describe, investigate, simulate, and forecast the response of the human immune system to a personalized cancer vaccine, our research group has developed a mechanistic mathematical model. This model incorporates various inputs, such as the vaccine that includes tumor-specific antigen peptides and adjuvant, the patient’s major histocompatibility complex (MHCs) I and II copy numbers, tumor size, T cells, and antigen-presenting cells [1].
Our mechanistic model consists of a system of ordinary differential equations, which effectively represent the cascade of immune responses triggered by the vaccine. Despite its comprehensiveness, the model presents challenges for mathematical analysis. Notably, some components of the model rely on phenomenological formulations, such as the representation of cancer cells elimination by T cells. Due to the limitations of the available data and our current understanding, determining the optimal approach for phenomenological modeling in this context remains inconclusive.
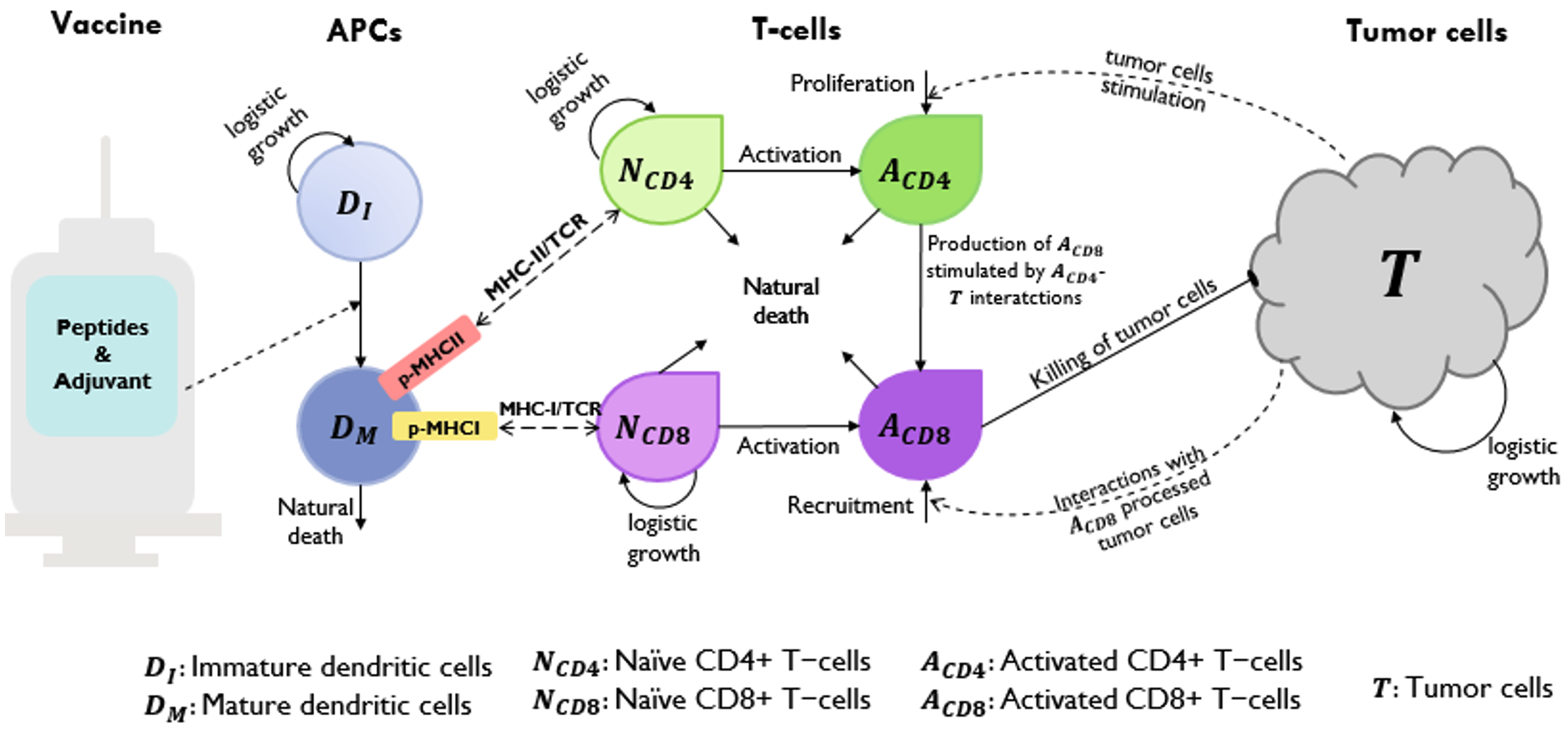
To increase the efficiency of the analysis, we simplified our previous model to three differential equations that describe the dynamics of vaccine peptides, cancer cells, and T cells [2]. A quasi-steady state approximation makes this simplified model amenable to phase plane analysis, a classical and powerful technique for analyzing planar dynamical systems. We focused on two popular forms of killing rate functions: the law of mass action (LMA) and the dePillis-Radunskaya Law (LPR) [3]. Using LMA, our results reveal that a slowly growing tumor can escape immune surveillance, a surprising but well-known phenomenon in cancer biology. The LPR offers richer dynamics, in which tumor elimination and uncontrolled tumor growth are both present. We showed that tumor elimination modeled by LPR requires sufficient initial activation of T cells in response to the malignant cells, which lends support to using the neoantigen cancer vaccine as an adjuvant therapy. In some sense, both LMA and LPR yield biologically or medically relevant interpretations and likely capture a certain level or aspect of clinical reality. Our work highlights the potential discrepancies in the interpretation of alternative phenomenological formulations in mathematical models. This can be an issue when the models are intended for medical use.
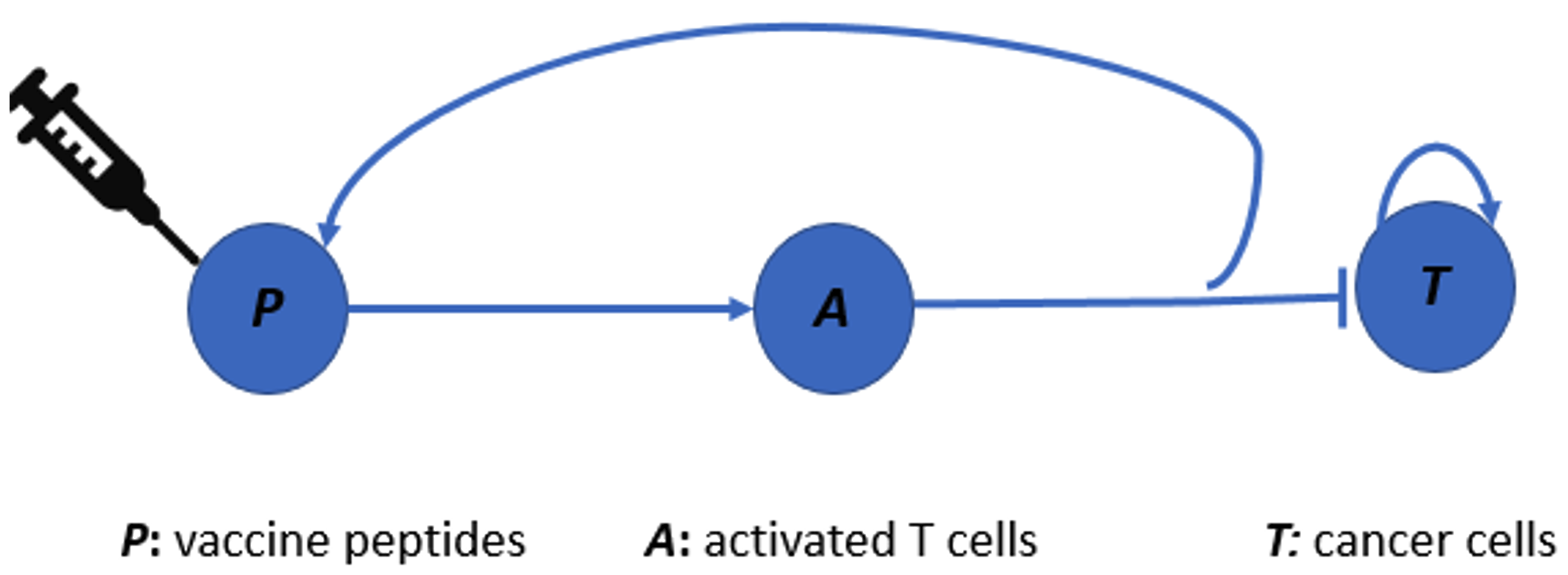
The process of mathematical modeling for neoantigen cancer vaccines, like any other modeling endeavor, is dynamic and iterative. Through incremental improvements based on our growing understanding of the treatment landscape, we aim to develop a more refined model that can eventually be applied in practical settings.
Currently, our focus lies in utilizing optimal control theory to enhance our original model. Specifically, we are leveraging this theory to optimize the dose of personalized cancer vaccines for individual patients within a fixed vaccination schedule. We have formulated two distinct minimization problems for this process (which can be thought of as clinical/treatment targets). The first problem aims to minimize tumor size, while the second problem incorporates an additional consideration of minimizing the excess of T cells alongside tumor reduction. Notably, our objective in both cases is to achieve the lowest possible vaccine dose that minimizes either problem.
The findings of our study highlight a consistent pattern in the optimal vaccine dosing strategy. Irrespective of the specific minimization problem considered, our results demonstrate that optimal vaccination strategies tend to involve higher doses during early vaccinations, followed by lower doses in subsequent treatment stages. These insights contribute to advancing our understanding of the optimal administration of neoantigen cancer vaccines.
Modeling Chimeric Antigen Receptor (CAR) T Cell Therapies
CAR T cell therapies have been remarkably successful, especially for advanced hematological malignancies, with six FDA-approved CAR T cell therapies currently available. Furthermore, research toward using CAR T cell therapy to treat other kinds of tumors, including solid tumors, is rapidly moving forward. Though CAR T cell therapies have shown tremendous success against hematological malignancies with good clinical responses, there are still some uncertainties, including antigen escape, therapeutic effect wear-off, and limitations directly related to CAR T cell activity, including adverse events like cytokine release syndrome (CRS) and neurotoxicity [5].
A clear understanding of the factors responsible for varying degrees of response in patients is necessary for refining patient selection to achieve increased efficacy.
Extraordinary response rates in B-cell leukemias were evidenced, but only a small subset of the CLL patients showed response to CAR T cell therapy.
We developed a mechanistic mathematical model to identify these factors and explore interactions between CAR T cells, inflammatory cytokines, and patients' tumor burden. The proposed model is a system of coupled ordinary differential equations designed based on well-known immunological principles and widely accepted views on the mechanism of CAR T cell kinetics, IL-6 secretion, and tumor killing in CAR T cell therapy.
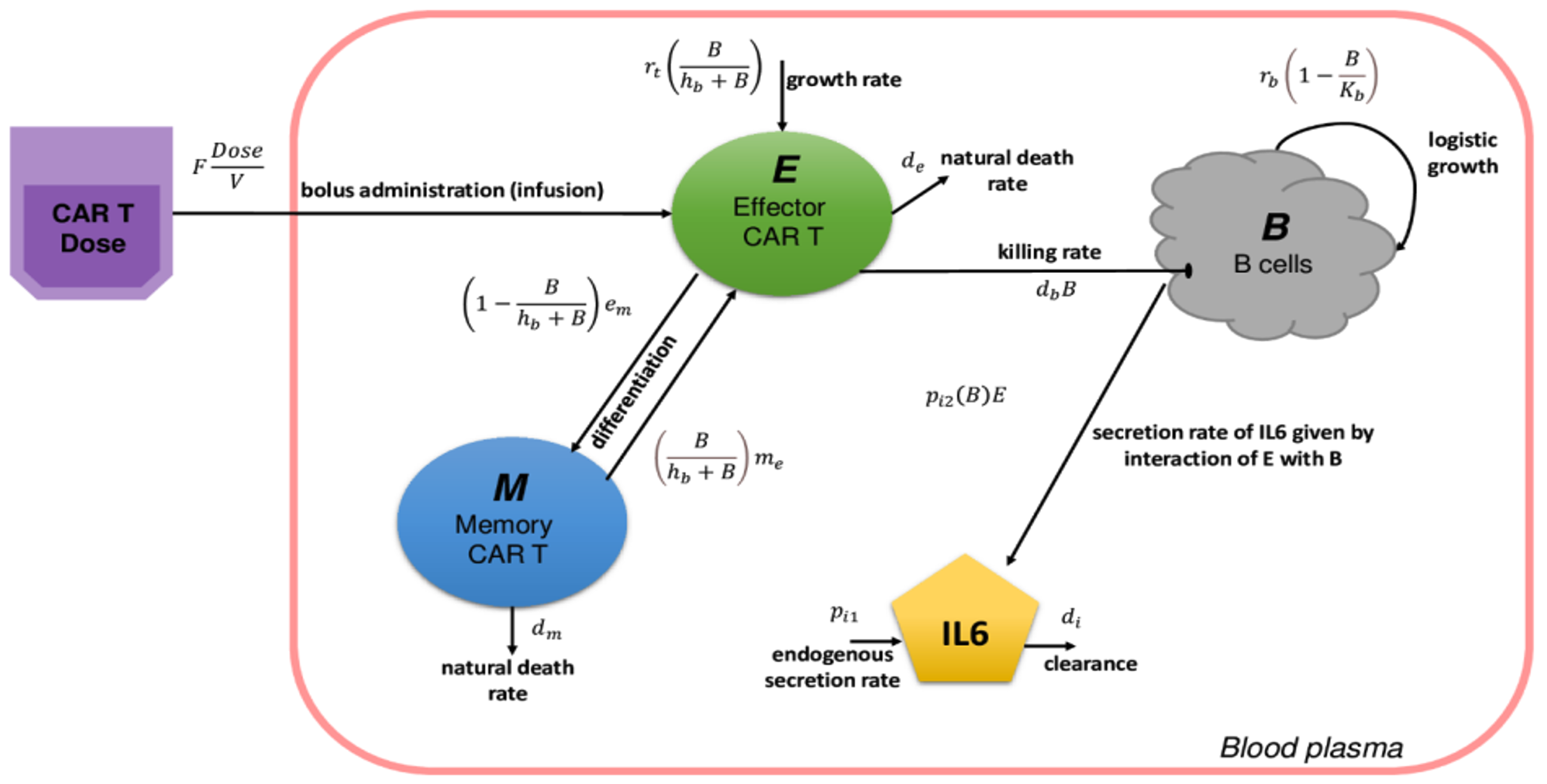
This model reports in silico disease outcomes in terms of B cell concentration, and these results are consistent with the in vitro experimental observations that CAR T cell characteristics, such as tumor cell killing capacity and T cell proliferation rate playing an important role in the patient response. We demonstrate the utility of mathematical modeling in understanding the factors that play an important role in patient response to CAR-T cell therapy. These results have been put into a manuscript [6], and we anticipate publishing it soon. This model may be further developed into a more comprehensive model to study CAR T cell therapies for other types of cancer.
Disclaimer
Our comments and contributions are an informal communication and represent our own best judgment. These comments do not bind or obligate FDA.
References
- Rodriguez Messan, M., Yogurtcu, O. N., McGill, J. R., Nukala, U., Sauna, Z. E., & Yang, H. (2021). Mathematical model of a personalized neoantigen cancer vaccine and the human immune system. PLoS Computational Biology, 17(9), e1009318.
- Han, L., Messan, M. R., Yogurtcu, O. N., Nukala, U., & Yang, H. (2023). Analysis of tumor-immune functional responses in a mathematical model of neoantigen cancer vaccines. Mathematical Biosciences, 108966.
- de Pillis, L. G., Radunskaya, A. E., & Wiseman, C. L. (2005). A validated mathematical model of cell-mediated immune response to tumor growth. Cancer research, 65(17), 7950-7958.
- Valega-Mackenzie, Wencel, et al. "Dose optimization of an adjuvanted peptide-based personalized neoantigen melanoma vaccine. " bioRxiv (2023): 2023-6.
- Nukala, Ujwani, et al. "A Systematic Review of the Efforts and Hindrances of Modeling and Simulation of CAR T-cell Therapy." The AAPS Journal 23 (2021): 1-20.
- Nukala, Ujwani, et al. "Modeling and Simulation of CAR T cell Therapy in Chronic Lymphocytic Leukemia Patients." medRxiv (2022): 2022-12.
© 2026 - The Mathematical Oncology Blog