Myeloid-derived suppressor cell dynamics control outcomes in the metastatic niche
Behind the Paper
Myeloid-derived suppressor cell dynamics control outcomes in the metastatic niche
Jesse Kreger, Evanthia T. Roussos Torres, Adam L. MacLean
Read the preprint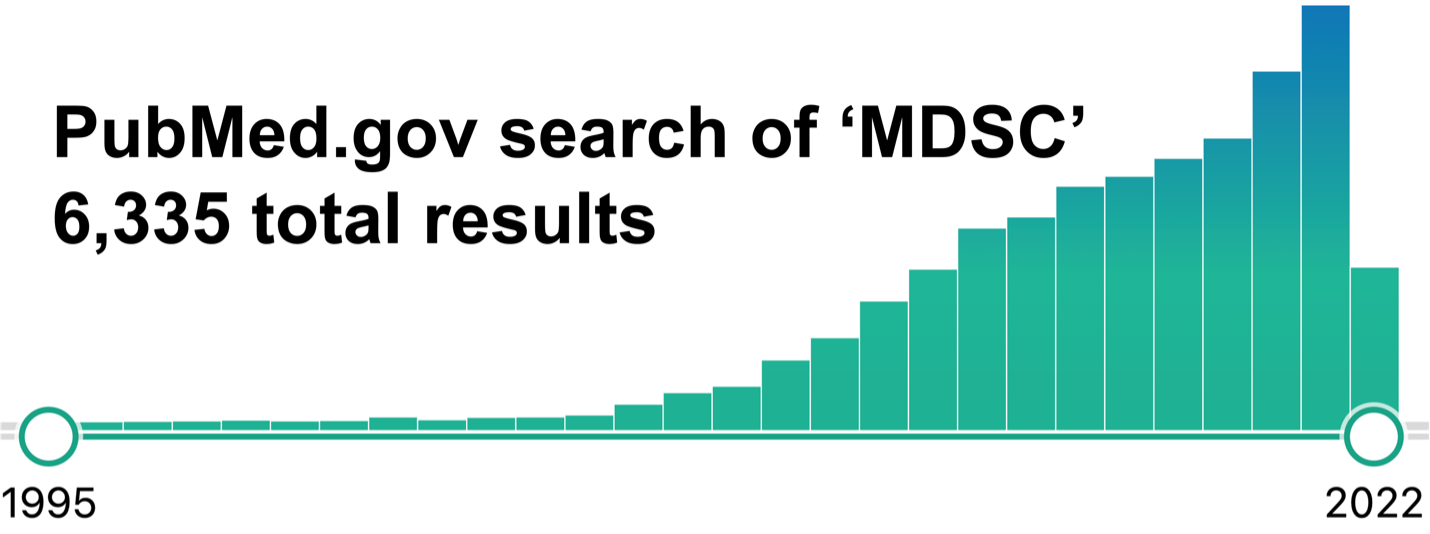
Figure 1: Number of papers on PubMed.gov when searching for 'MDSC’. The maximum to date is 1,046 papers in 2021.
Previous work led by Evanthia Roussos Torres’s lab uncovered a crucial role for MDSCs in HER2+ breast cancer5. In an ongoing collaboration between our labs (stay tuned, exciting things to come!), we have discovered that the influence of MDSCs in breast cancer metastases is potentially even larger. In this paper, we take a theoretical approach and present a feedback stochastic delay differential equation model of the tumor-immune-MDSC interactions occurring in and around the site of a new metastatic growth (Figure 2). We simulate the battle between the pro-tumor (cancer cells, MDSCs) and anti-tumor (NK and CTL) populations, and identify MDSC-focused strategies that shift the balance of power toward the anti-tumor populations6 – resulting in up to a 50% reduced probability of a successful metastasis forming.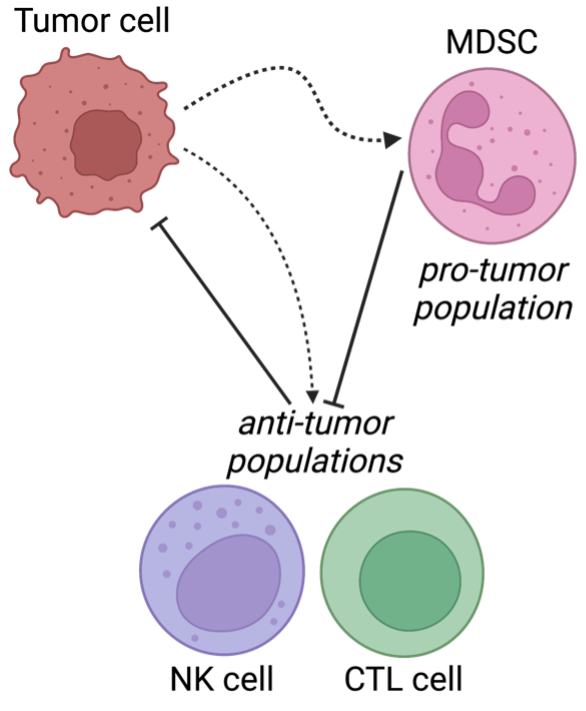
Figure 2: Targeting the sidekick instead of the tumor? Therapies that target MDSCs can be effective against tumor progression. Model dynamics: the MDSC population inhibits the NK and CTL populations, and the NK and CTL populations inhibit the tumor population.
Furthermore, fitting in vivo tumor data to our model using Bayesian parameter inference reveals that the strength of immune-suppressiveness – as controlled by NK cell inhibition by MDSCs – is the most important parameter in determining tumor outcome (Figure 3). This has direct clinical implications: while it may not yet be possible to directly modulate this parameter in a clinical setting, we highlight the potential importance of interventions targeting MDSCs. Moreover, the successful fitting of various tumor responses to tumor-MDSC dynamic models demonstrates our ability to build and fit patient-specific tumor growth models, with which to study metastatic dynamics in vivo. Understanding tumor-immune-MDSC dynamics is in its nature a systems biology problem: where mathematical modeling, computational algorithms, and experiments are essential to tease apart the intricate relationships involved. We have taken care to elucidate our assumptions and conclusions clearly, aiming for an audience including both mathematicians and clinicians. The anticipated impact of our work for cancer biologists is the identification of MDSC parameters as critical in determining metastatic outcomes, along with quantitative predictions of the best possible interventions in MDSC-rich environments. In addition, for computational biologists, we anticipate additional impacts to include:- a validated model of tumor-MDSC dynamics to simulate either deterministic or stochastic tumor growth, that can be fit to other tumor types given data
- a framework for parameter inference of tumor-immune dynamics via Bayesian inference using RECIST data to constrain the models.
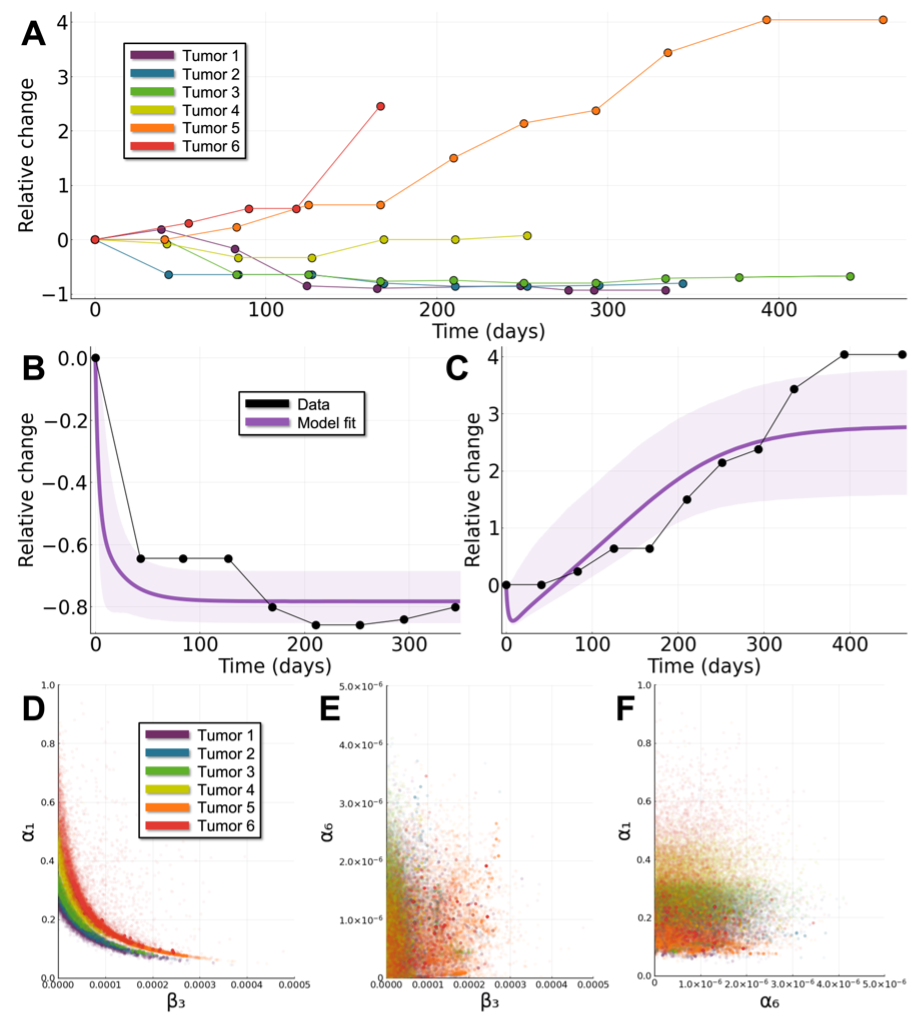
Figure 3: Interactions between MDSCs and NK cells control clinical tumor growth outcomes. A: Relative change in tumor size from the baseline assessment for six tumors undergoing treatment. B-C: Tumor and model trajectories based on the relative change in the tumor population. D-F: Samples from the posterior distribution from Bayesian parameter inference (D); NK cell inhibition rate by MDSCs (\(\beta_3\)) versus tumor growth rate (\(\alpha_1\)). (E); NK cell inhibition rate by MDSCs (\(\beta_3\)) versus CTL stimulation by tumor-NK cell interaction (\(\alpha_6\)) (F); CTL stimulation by tumor-NK cell interaction (\(\alpha_6\)) versus tumor growth rate (\(\alpha_1\)).
References
- Tesi, R. J. MDSC; the Most Important Cell You Have Never Heard Of. Trends Pharmacol. Sci. 40, 4–7 (2019).
- Veglia, F., Sanseviero, E. & Gabrilovich, D. I. Myeloid-derived suppressor cells in the era of increasing myeloid cell diversity. Nat. Rev. Immunol. 21, 485–498 (2021).
- Bergenfelz, C. & Leandersson, K. The Generation and Identity of Human Myeloid-Derived Suppressor Cells. Front. Oncol. 10, (2020).
- Markowitz, J., Wesolowski, R., Papenfuss, T., Brooks, T. R. & Carson, W. E. Myeloid-derived suppressor cells in breast cancer. Breast Cancer Res. Treat. 140, 13–21 (2013).
- Sidiropoulos, D. N. et al. Entinostat Decreases Immune Suppression to Promote Antitumor Responses in a HER2+ Breast Tumor Microenvironment. Cancer Immunol. Res. 10, 656–669 (2022).
- Kumar, V., Patel, S., Tcyganov, E. & Gabrilovich, D. I. The nature of myeloid-derived suppressor cells in the tumor microenvironment. Trends Immunol. 37, 208–220 (2016).
© 2026 - The Mathematical Oncology Blog