Adaptive Therapy in 2021
Math Modeling: Year in Review
The concept of adaptive therapy is exciting to many of us in mathematical oncology for at least three reasons: 1) this approach to therapy is based on evolutionary concepts, 2) this approach to therapy is informed by mathematical modeling, and 3) this approach has been tested in clinical trials. 2021 saw the release of important data from the trial, including Gleason scores, metastatic locations, initial PSA values, and cumulative dose of both the standard of care cohort and its adaptive counterpart1. It's likely that we may never seen a randomized clinical trial in this setting, due to the fact that standard of care for metastatic prostate cancer has changed with early abiraterone administration in the castrate sensitive setting1. (The trial administered abiraterone adaptively in the castrate-resistant setting.)
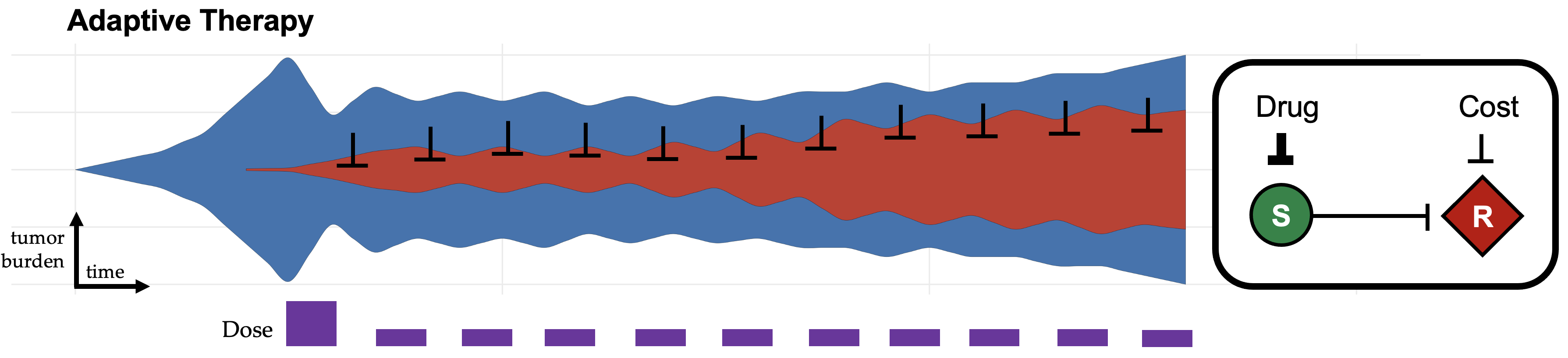 The paper also notes that the trial has inspired deeper investigations into the mathematical models of adaptive therapy to investigate the clinical circumstances when adaptive approaches are likely to be effective. We'll explore some of these approaches that were published in 2021, below (listed in no particular order).
The paper also notes that the trial has inspired deeper investigations into the mathematical models of adaptive therapy to investigate the clinical circumstances when adaptive approaches are likely to be effective. We'll explore some of these approaches that were published in 2021, below (listed in no particular order).
Published in 2021
Darwinian Approaches for Cancer Treatment: Benefits of Mathematical Modeling
Sophia Belkhir, Frederic Thomas, Benjamin Roche
Read the paperDo mechanisms matter? Comparing cancer treatment strategies across mathematical models and outcome objectives
Cassidy K. Buhler, Rebecca S. Terry, Kathryn G. Link, Frederick R. Adler
Read the paperPredicting patient-specific response to adaptive therapy in metastatic castration-resistant prostate cancer using prostate-specific antigen dynamics
Renee Brady-Nicholls, Jingsong Zhang, Tian Zhang, Andrew Z. Wang, Robert Butler, Robert A. Gatenby, Heiko Enderling
Read the paperOptimizing Adaptive Therapy Based on the Reachability to Tumor Resistant Subpopulation
Jiali Wang, Yixuan Zhang, Xiaoquan Liu, Haochen Liu
Read the paperIs the Fixed Periodic Treatment Effective for the Tumor System without Complete Information?
Jiali Wang, Yixuan Zhang, Xiaoquan Liu, Haochen Liu
Read the paperAre Adaptive Chemotherapy Schedules Robust? A Three-Strategy Stochastic Evolutionary Game Theory Model
Rajvir Dua, Yongqian Ma, Paul K. Newton
Read the paperPreprints published in 2021
Where would science be without preprints? Let's highlight a few preprints from 2021.Spatial structure impacts adaptive therapy by shaping intra-tumoral competition
Maximilian A. R. Strobl, Jill Gallaher, Jeffrey West, Mark Robertson-Tessi, Philip K. Maini, Alexander R. A. Anderson
Read the preprintThe impact of the spatial heterogeneity of resistant cells and fibroblasts on treatment response
Masud M A, Jae-Young Kim, Cheol-Ho Pan, Eunjung Kim
Read the preprintThe impact of the spatial heterogeneity of resistant cells and fibroblasts on treatment response
Masud M A, Jae-Young Kim, Cheol-Ho Pan, Eunjung Kim
Read the preprintLooking forward in 2022
I would be remiss not to close out this post without looking forward to the yet unanswered questions surrounding adaptive therapy. I hope 2022 will bring more mathematical models to investigate the following topics:- What diseases are ideal targets for adaptive therapy?
- What is the role of plasticity in adaptive treatment?
- What are the best practices to extending adaptive therapy to multiple drugs?
- Can mathematical modeling drive personalized treatment scheduling in the clinic?
References
- Zhang, J., Cunningham, J.J., Brown, J.S. et al. Response to Mistry. Nat Commun 12, 329 (2021). https://doi.org/10.1038/s41467-020-20175-3
- Gatenby, Robert A., et al. "Adaptive therapy." Cancer Research 69.11 (2009): 4894-4903.
- Belkhir, Sophia, Frederic Thomas, and Benjamin Roche. "Darwinian approaches for cancer treatment: benefits of mathematical modeling." Cancers 13.17 (2021): 4448.
- Buhler, Cassidy K., et al. "Do mechanisms matter? Comparing cancer treatment strategies across mathematical models and outcome objectives." Mathematical Biosciences and Engineering 18.5 (2021): 6305-6327.
- Viossat, Yannick, and Robert Noble. "A theoretical analysis of tumour containment." Nature Ecology & Evolution 5.6 (2021): 826-835.
- Brady-Nicholls, Renee, et al. "Predicting patient-specific response to adaptive therapy in metastatic castration-resistant prostate cancer using prostate-specific antigen dynamics." Neoplasia 23.9 (2021): 851-858.
- Wang, Jiali, et al. "Optimizing adaptive therapy based on the reachability to tumor resistant subpopulation." Cancers 13.21 (2021): 5262.
- Wang, Jiali, et al. "Is the Fixed Periodic Treatment Effective for the Tumor System without Complete Information?." Cancer Management and Research 13 (2021): 8915.
- West, Jeffrey, et al. "Towards multidrug adaptive therapy." Cancer Research 80.7 (2020): 1578-1589.
- Dua, Rajvir, Yongqian Ma, and Paul K. Newton. "Are adaptive chemotherapy schedules robust? A three-strategy stochastic evolutionary game theory model." Cancers 13.12 (2021): 2880.
- Strobl, Maximilian AR, et al. "Turnover modulates the need for a cost of resistance in adaptive therapy." Cancer Research 81.4 (2021): 1135-1147.
- Strobl, Maximilian AR, et al. "Spatial structure impacts adaptive therapy by shaping intra-tumoral competition." bioRxiv (2021): 2020-11.
- Masud, M. A., et al. "The impact of the spatial heterogeneity of resistant cells and fibroblasts on treatment response." bioRxiv (2021).
© 2026 - The Mathematical Oncology Blog