Bridging the gap between theory and clinic: the importance of measuring pairwise interactions
Behind the paper
Measuring competitive exclusion in non-small cell lung cancer
Nathan Farrokhian, Jeff Maltas, Mina Dinh, Arda Durmaz, Patrick Ellsworth, Masahiro Hitomi, Erin McClure, Andriy Marusyk, Artem Kaznatcheev, Jacob G Scott
Read the preprint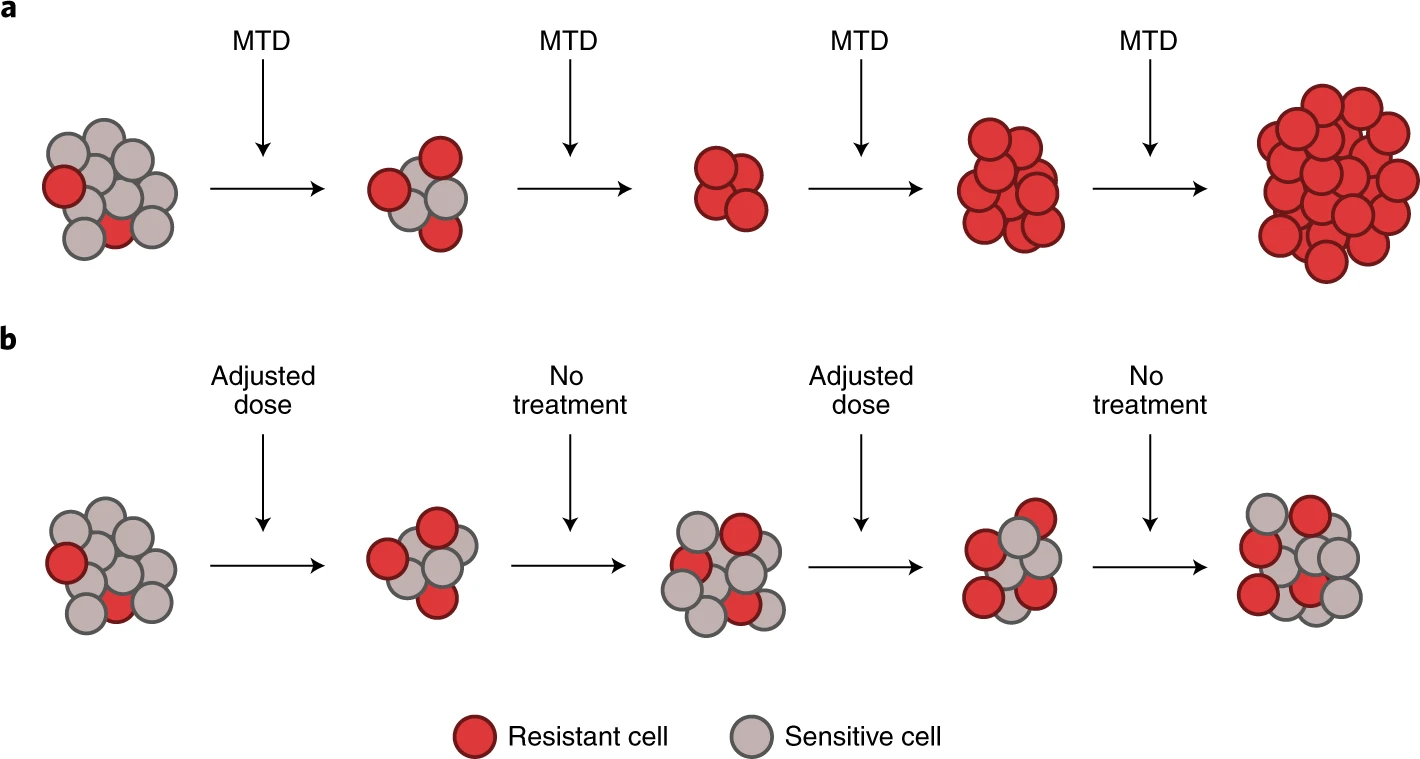
Figure 1
To understand this outcome, it is important to realize that tumor subclones do not live or reproduce in isolation. Genomic instability inherent to the mechanism of oncogenesis promotes intratumoral heterogeneity. With this diverse cellular population comes a diverse range of intercellular interactions. These dynamics are thought to drive resulting equilibrium amongst the constituents. As such mathematical modeling of these interactions with game theoretical methodology has gained in popularity. Dynamic therapeutic protocols using models of this type have already made their way into the clinic with promising results (Figure 2). Specifically, a clinical trial in castration resistant prostate cancer utilized this framework to develop an adaptive protocol that not only decreased cumulative drug dose, but more importantly significantly lengthened time to progression in this population.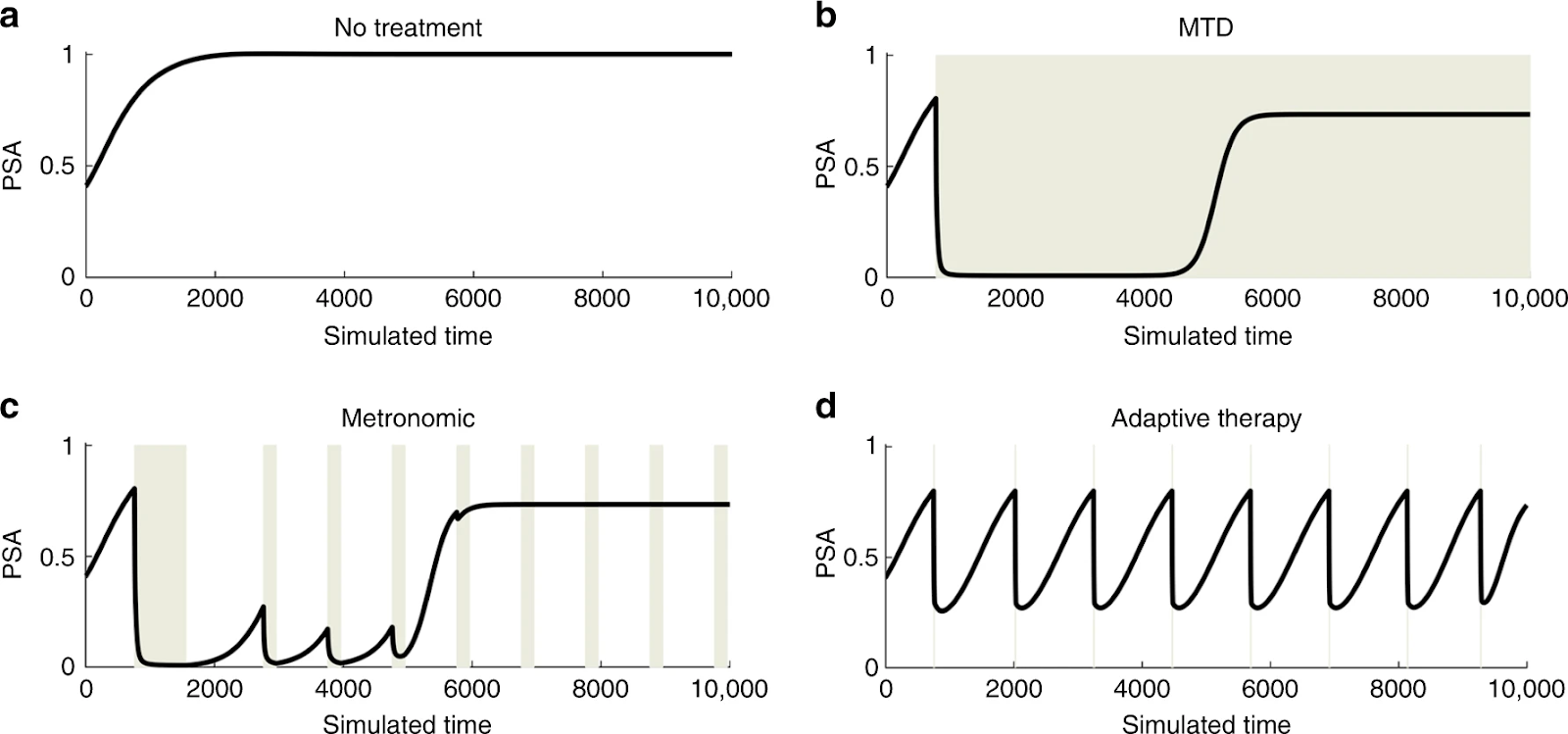
Figure 2
While these successes highlight the potential of these models to revolutionize chemotherapeutic protocols, they often rely heavily on biological conjecture and intuition to derive model parameters. These parameters drive the resulting evolutionary equilibria and even small changes in their relative values can result in completely different ecological endpoints (Figure 3).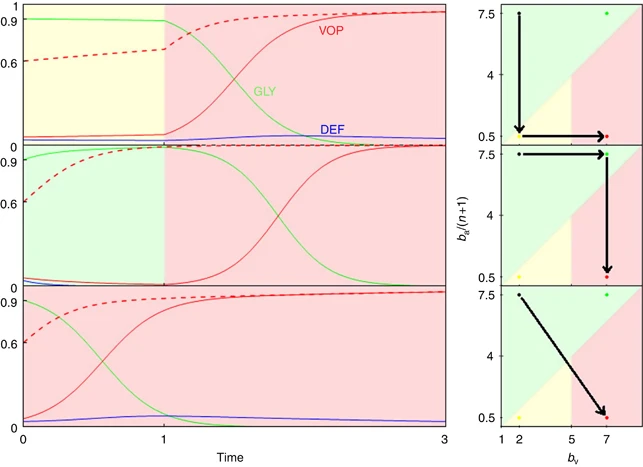
Figure 3
To bridge this gap between theory and clinical application, Kaznatcheev et al. developed a method to measure and parametrize these games and their evolutionary consequences in the in vitro setting (Figure 4).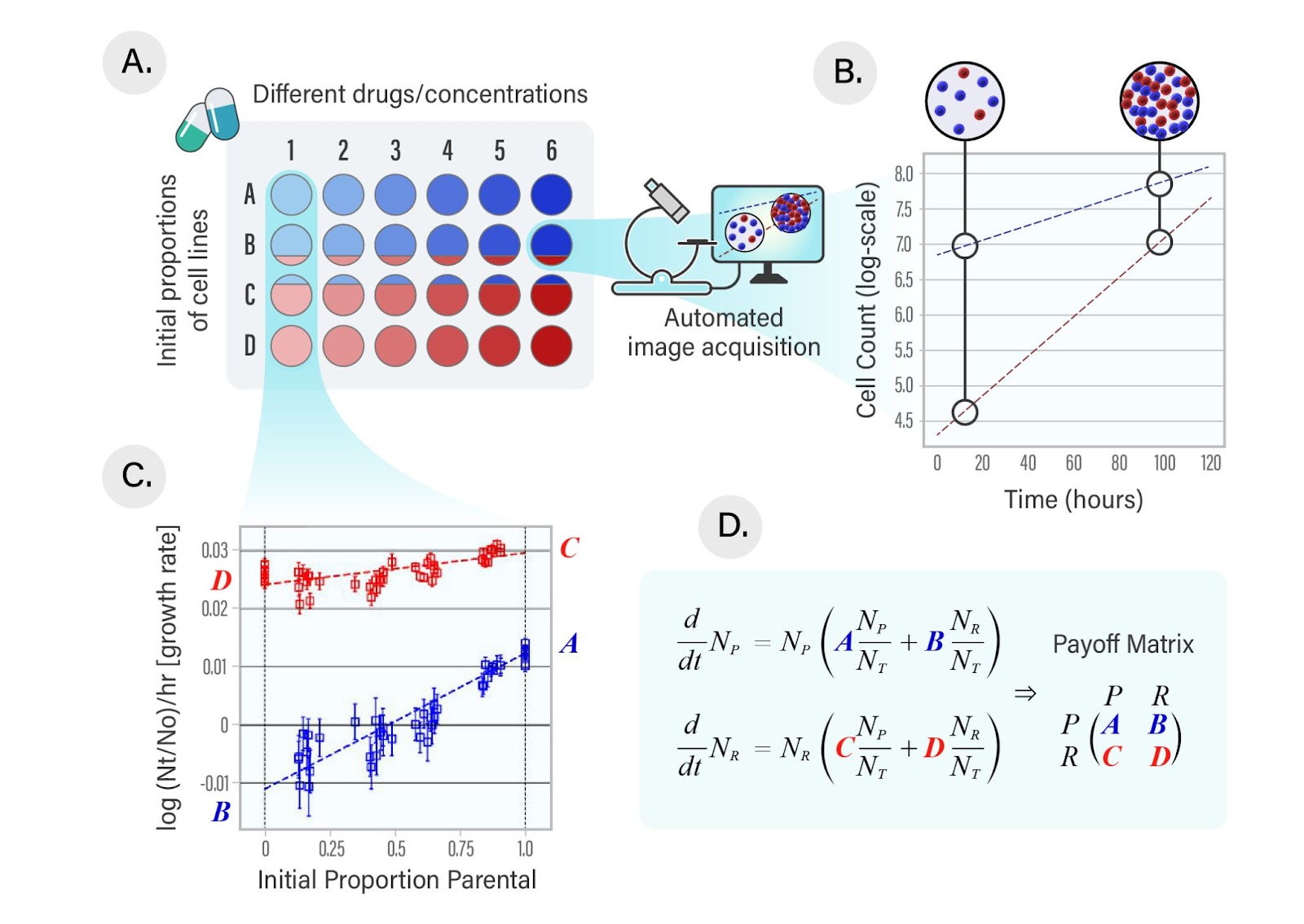
Figure 4
In our recent preprint, we utilized this methodology to show that frequency dependent interactions ameliorate the fitness cost of gefitinib resistance in an in vitro EGFR+ lung cancer population, which leads to a potential safe harbor for small resistant sub-populations (Figure 5).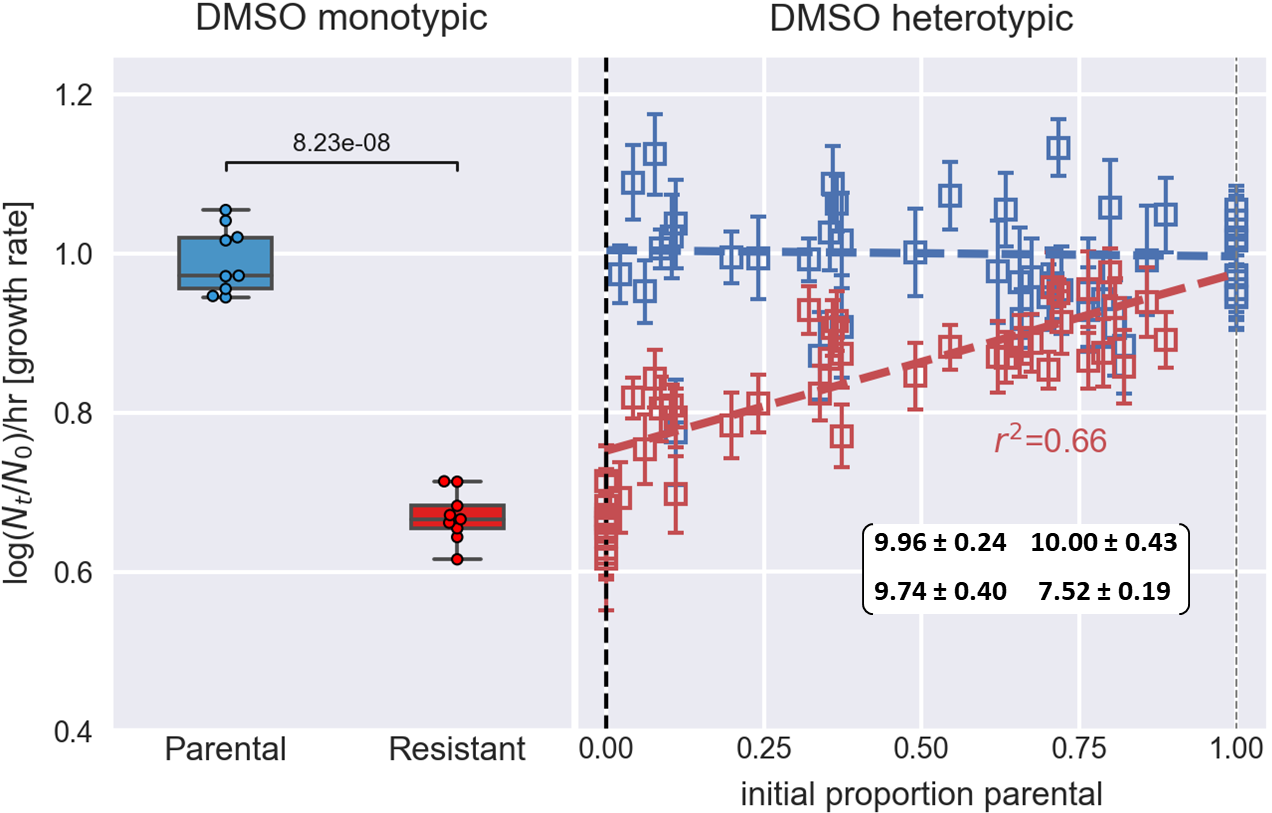
Figure 5
As expected, introduction of gefitinib reverses these interactions such that the resistant population promotes more rapid extinction of the non-resistant cells. Most importantly, at clinically relevant doses, this interaction outweighs the effect of the drug itself (Figure 6).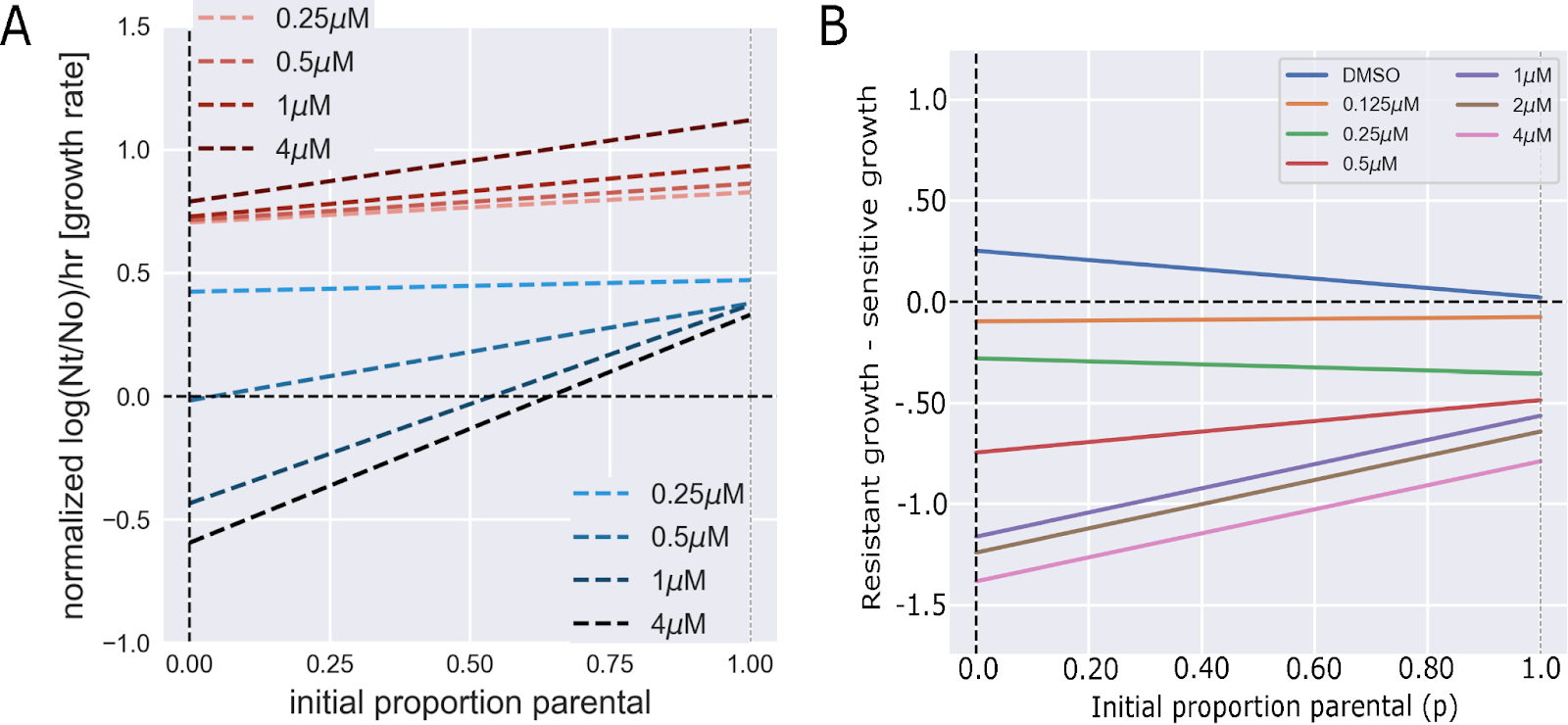
Figure 6
Using these in vitro derived parameters, we generated in silico models to investigate the implication of these frequency dependent interactions (Figure 7). As expected, higher doses of gefitinib resulted in larger initial decreases in tumor volume. This was largely due to accelerated extinction of the parental cells driven by interaction with the resistant cells. Importantly, this rapid extinction allowed for more rapid expansion of the resistant cells. As a result, this rapid reduction in tumor volume was met with a similarly rapid rebound in disease burden. Comparatively, while lower doses of gefitinib resulted in more modest initial decreases in tumor volume, maintenance of underlying tumor heterogeneity promoted a more durable response (Figure 7E)..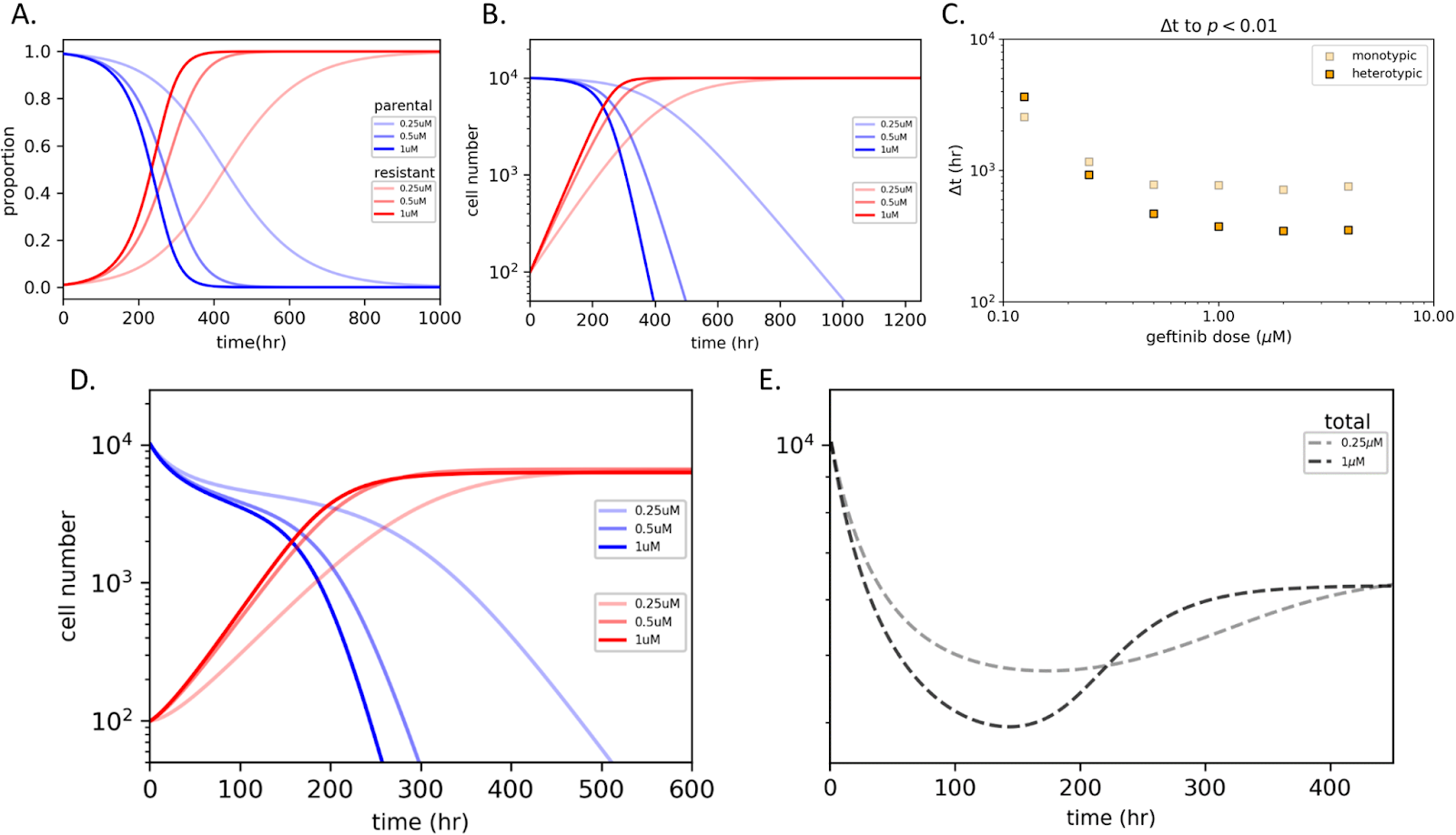
Figure 7
In other words, exploitation of the growth exclusion that results from interspecific competition allowed for improved chronic control of the tumor. This was largely dictated by the slower extinction of the parental population, whose presence continued to limit outgrowth of the resistant population - a feature that persisted across all models tested. As such, using monoculture estimates to predict shifts in population equilibria can be largely inaccurate and must be measured prior to the development of adaptive therapeutic protocols that rely on maintenance of underlying tumor heterogeneity.References
- Staňková, K. (2019). Resistance games. Nature ecology & evolution, 3(3), 336-337.
- Zhang, J., Cunningham, J. J., Brown, J. S., & Gatenby, R. A. (2017). Integrating evolutionary dynamics into treatment of metastatic castrate-resistant prostate cancer. Nature communications, 8(1), 1-9.
- Kaznatcheev, A., Vander Velde, R., Scott, J. G., & Basanta, D. (2017). Cancer treatment scheduling and dynamic heterogeneity in social dilemmas of tumour acidity and vasculature. British journal of cancer, 116(6), 785-792.
- Kaznatcheev, A., Peacock, J., Basanta, D., Marusyk, A., & Scott, J. G. (2019). Fibroblasts and alectinib switch the evolutionary games played by non-small cell lung cancer. Nature ecology & evolution, 3(3), 450-456.
- Farrokhian, N., Maltas, J., Dinh, M., Durmaz, A., Ellsworth, P., Hitomi, M., ... & Scott, J. G. (2020). Measuring competitive exclusion in non-small cell lung cancer. bioRxiv, 2020-09.
© 2026 - The Mathematical Oncology Blog