The Eye of the Needle in Oncolytic Virotherapy
What a cool idea: simply infect all cancer cells with a deadly virus, which then kills all cancer cells and problem solved! Wow. How did somebody come up with this thrilling idea? Well, it was observed in cancer patients who caught a viral infection that, sometimes, the tumor became smaller as a result of the infection. Since these observations, the hunt for the proper virus has started. Researchers look at herpes virus, measles virus, vaccina virus, vesicular stomatitis virus, adenoviruses and reoviruses, and probably more. Do they work? Well, two of these have already been approved by governmental health agencies, one is the adenovirus H101 for the treatment of head and neck cancer and another one is indeed a herpes virus (T-VEC) for the treatment of advanced melanoma.
However, oncolytic virotherapy has not fulfilled the expectations that people originally had. In some medical trials only 20% of patients responded. The problem is, of course, complicated. This is why mathematical modelling enters the discussion. In a patient, a viral infection has (at least) two effects. Firstly, it infects the cells, in this case the cancer cells, and kills a certain number due to viral reproduction and subsequent cell lysis. However, it is also an infection and the immune response is not going to sit idle and watch. After a few days the adaptive immune response is starting to destroy infected cancer cells. That’s good, but if this happens too fast, then the viral infection is terminated before it reaches all the cancer cells in the body. There is a tight time window in which the viral infection needs to be efficient and the immune response slow, otherwise the viral infection is stopped before it fulfilled its task. I call this effect the eye of the needle in oncolytic virothepary. Its mechanisms have been identified through mathematical modelling.
In fact, the mathematical modelling of oncolytic virotherapy is a rich and interesting subject. If you are interested to learn more, I recommend to start with the papers of Wodarz [5], Pooladvand [3], Jenner [2], Storey [4], and our own recent submission [1]. In particular in [1] we give a comprehensive review of existing modelling. A common theme arises, as many authors find that, for successful treatment, the virus has to spread fast and reproduce large amounts of virions (large burst sizes), not surprising.
The basic model for oncolytic viruses is essentially an SIV predator-prey model, i.e., susceptible cancer cells, infected cancer cells and the virus as predator. As such, the system has a Hopf bifurcation and periodic solutions. These periodic solutions have been analysed in great detail, including more cell compartments, time delays, immune responses and spatial dependence. The spatial dependence is actually rather interesting. As the basic SIV model is oscillatory, a spatial version corresponds to coupled spatial oscillators. We know, for example through Steven Strogatz’ work, that coupled oscillators can form all kinds of spatial patterns, from hollow rings to spirals to chaotic dynamics. In the ideal case, we want to infect the tumor uniformly, with virus particles everywhere, and that before the immune response wipes everything out. However, due to the time scales involved, spatial heterogeneities might arise. Wodarz et al. [5] suggested that in those cases the hollow ring pattern is the most advantageous. A virus wave travels over the domain and leaves dead cancer cells behind. We confirm this with our PDE model in [1] and I show a simulation here, which was made by my PhD student Arwa Baabdulla. Here C shows the cancer cells (brown is low, black is high), I the infected cancer cells (blue is low and white is high), and V the virus (blue is low and red is high). This ring travels outward with a constant velocity.
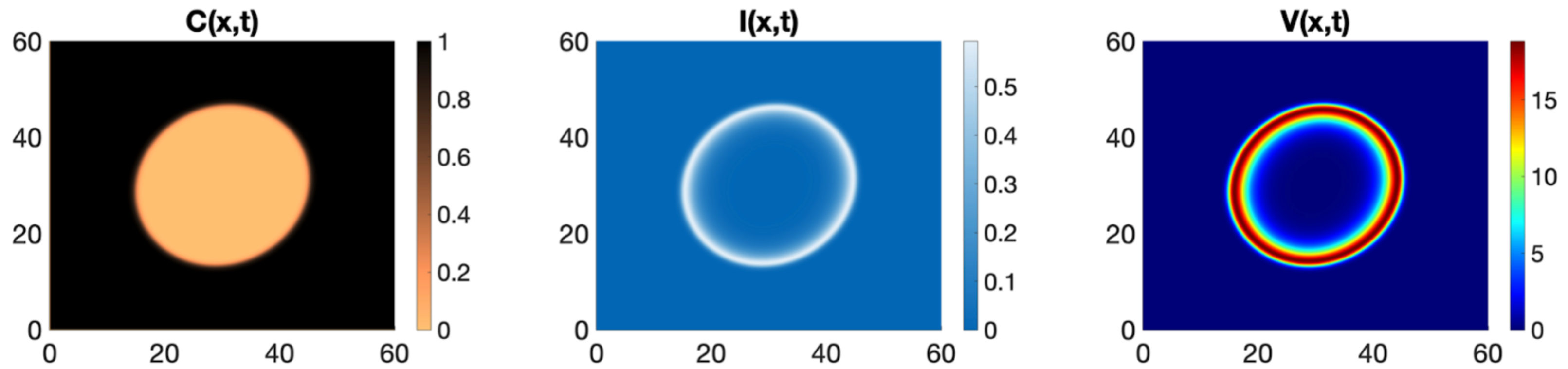
If the simulations are run longer, we see that the cancer comes back in the center of the domain, followed by virus outburst and another ring starts to follow the first ring. We find a spatially heterogeneous periodic solution. However, between the oscillations, the cancer cell density gets so low, that it can be considered to be removed. In [1] we make this formal by using the tumor control probability, which is essentially equal to 1 between the oscillation cycles. By further increasing the viral efficacy in our model, we also find very complex spatio-temporal dynamics of spiral waves and irregular oscillations. Let me show a beautiful example here, where we thank Andrew Krause and his team for making VisualPDE freely available.
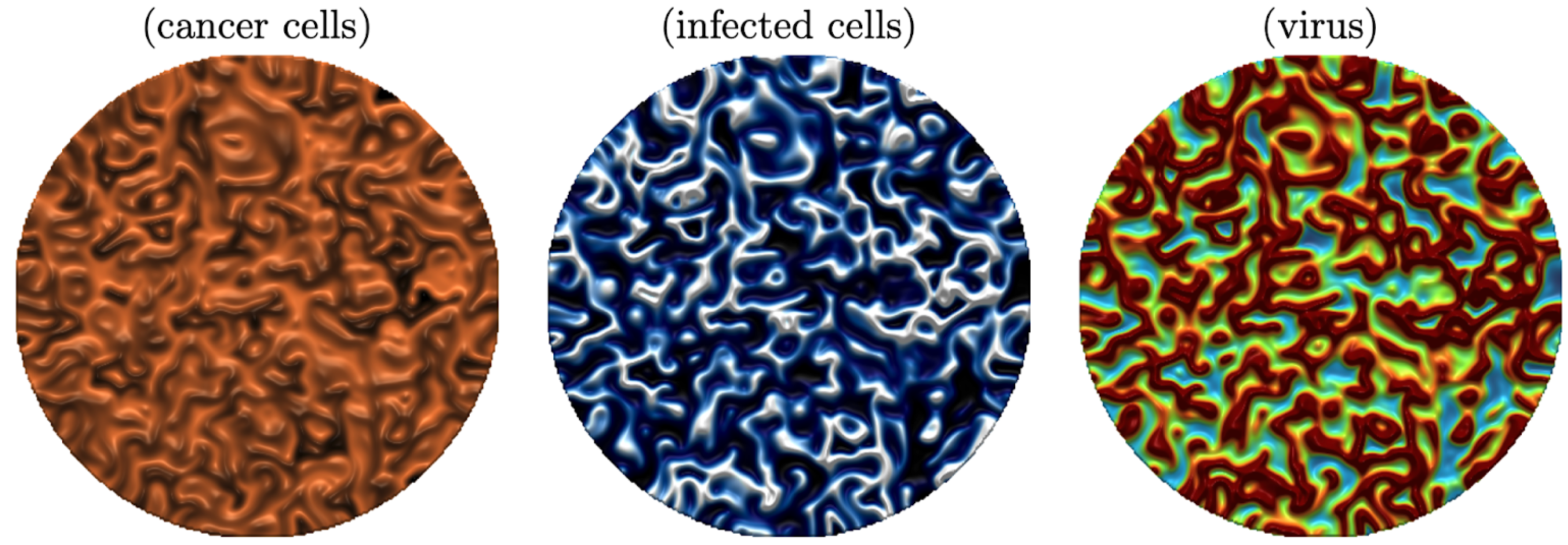
We see that the modelling of oncolytic virotherapy can lead to very interesting mathematical questions. However, the real question is how to make it more efficient in a real application. As the window of opportunity is only a few days long, scientists consider ideas to either make the virus more efficient, or delay the immune response, or to make the immune response more (or less) efficient. All this can be achieved through combination of viral therapy with other therapies such as radiation, chemotherapy, immunotherapy or even a second virus. Very interesting models have been considered by several authors, and we give a comprehensive list of references in [1]. Yet, so far we did not find a good way to open the eye of the needle in oncolytic virotherapy in a consistent and efficient way. Many more interesting questions are still unanswered.
References
- Arwa Abdulla Baabdulla, Thomas Hillen, Oscillations in a Spatial Oncolytic Virus Model. BioRxiv doi: 10.1101/2023.12.19.572433
- Adrianne L Jenner, Chae-Ok Yun, Peter S Kim, and Adelle CF Coster. Mathematical modelling of the interaction between cancer cells and an oncolytic virus: insights into the effects of treatment protocols. Bulletin of mathematical biology, 80(6):1615–1629, 2018.
- P. Pooladvand, CO. Yun, AR. Yoon, PS. Kim, and F. Frascoli. The role of viral infectivity in oncolytic virotherapy outcomes: A mathematical study. Mathematical Biosciences, 334:108520, 2021.
- Kathleen M Storey, Sean E Lawler, and Trachette L Jackson. Modeling oncolytic viral therapy, immune checkpoint inhibition, and the complex dynamics of innate and adaptive immunity in glioblastoma treatment. Frontiers in physiology, 11:151, 2020.
- D. Wodarz, A. Hofacre, JW. Lau, Z. Sun, H. Fan, and NL. Komarova. Complex spatial dynamics of oncolytic viruses in vitro: mathematical and experimental approaches. PLoS Computational Biology, 8(6):e1002547, 2012.
Photo Credits
Title Photo by Gustavo Sánchez on Unsplash.© 2026 - The Mathematical Oncology Blog