Rethinking Angiogenesis
Behind the paper
Superlinear growth reveals the Allee effect in tumors
Youness Azimzade, Abbas Ali Saberi, Robert A. Gatenby
Read the paperWhat are the dynamics of angiogenesis during tumor growth? Are tumors well-vascularized after successful angiogenetic switch at early stages or are they continuously struggling to push the oxygen supply limit to grow larger? The majority of existing literature suggests that small tumors normally experience oxygen shortage. As they become successful in inducing angiogenesis (a discrete step known as angiogenetic switch1) nutrient shortage becomes of less importance, at least for cells living in tumor border. However, you might be surprised to know that one of the greatest figures in tumor angiogenesis research, Judah Folkman, and his colleagues had a different perspective. In a well-cited paper2, they suggested that tumors live in an environment with limited nutrient supply that imposes a carrying capacity. Tumors need to produce angiogenetic signals to push this limit further to grow larger. If tumors fail to induce more angiogenesis, they face postvascular dormancy. Once tumors escape such a dormancy, they grow exponentially3. However, such a dynamical understanding of angiogenesis and postvascular dormancy has gained little attention so far, possibly due to a lack of concrete clinical supporting data. If clinical data shows exponential growth for at least a fraction of tumors, then we can conclude that those tumors have previously experienced postvascular dormancy and have now escaped it. Recently, it was found that for a variety of human tumors, growth rate is super-linearly scaled by tumor size as \(\frac{dC_T}{dt} \propto C_T^{\beta}\) with \(\beta=1.25\).4 A naïve interpretation of this data may suggest that these tumors are growing faster than exponentially growing populations, but they are not. In fact, this data reveals that tumor growth rate accelerates as it becomes larger, but we believe the driving mechanism was not identified properly. Using a model that incorporates internal competition among tumor cells and interactions with the environmental limitations, we show that the strange super-linear scaling results from tumors’ successful effort to induce further angiogenesis5. Based on these results, tumors continuously fight to induce more angiogenesis and push their limits forward to grow larger. Tumors induce more vessels and their growth exhibits super-linear dynamics. Under this understanding, angiogenesis is a dynamic process and acquiring necessary signals is just the beginning part of successful angiogenesis. In other words, super-linear growth is a result of accelerated angiogenesis and it emerges once tumors escape postvascular dormancy. Thus, the super-linear growth that has been observed across different solid tumors, is empirical evidence for postvascular dormancy that tumors experience and escape.
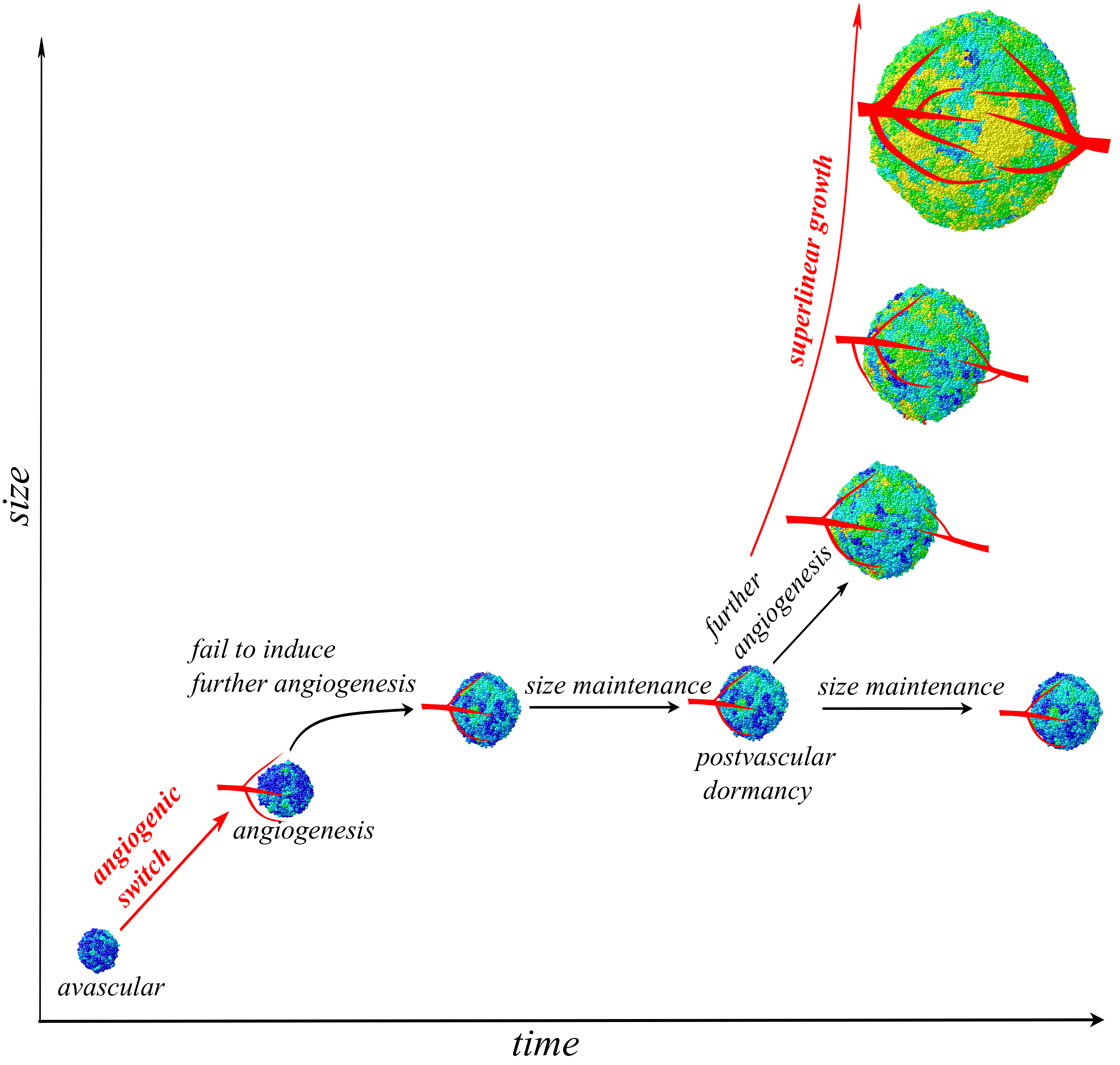
Figure 1: Super-linear growth emerges when previously starving tumors successfully induce further angiogenesis to grow larger.
We did a thorough analysis to check if alternative explanations such as evolutionary dynamics of competition between different sub-populations can lead to superlinear growth. However, our results suggest that such changes in growth rate are far too small to have a relevant effect on growth, a finding in line with previous results in the literature6. The study also highlights the role of Allee effects in cancer growth. Darwin was the first to note a puzzling phenomenon – he expected proliferation would decline as a population increased in size and approached its carrying capacity but observed exactly the opposite as proliferation often accelerated as the population grew. Warder Clyde Allee further studied this eponymous effect in detail, showing that the fitness of an individual within a population often increases as the population size grows due to the protective effects of large groups. Cancer cells, like all living systems, are subject to Darwinian dynamics but the emphasis is often on individual cancer cells as the unit of selection. Similarly, the role of angiogenesis in cancer biology has been extensively studied and anti-angiogenic treatments are frequently used in cancer therapy. Here, we point out that angiogenesis causes an Allee effect in the context of cancer evolution. Loosely organized groups of cancer cells must collectively produce sufficient angiogenic signals to promote vascular in-growth and, thus, increase their fitness. This is an example of niche construction or niche engineering. But, as always, evolution provides additional layers of complexity. Within each group of angiogenic cancer cells, there is an opportunity for “cheating”. Cells are producing a “common good” in the form of pro-angiogenic signals. But, as in the “tragedy of the commons,” some members of the group may adopt an alternative strategy that reduces or eliminates its production of angiogenic signals. Here, the individual cell benefits from the common good while not assuming the phenotypic cost of producing and excreting the signals. Ironically, this increases their fitness compared to their neighbors. But, of course, if too many cells in the group adopt this strategy, the niche construction project will fail. Although cancer cells are angiogenic, blood flow in tumors almost always exhibits striking spatial and temporal heterogeneity. These dynamics, which are critical factors in failure of cancer therapy, reflect the complex evolutionary interactions of individual cells and groups of cells that govern intra-tumoral niche construction.References
- Bergers, G. and Benjamin, L.E., 2003. Tumorigenesis and the angiogenic switch. Nature Reviews Cancer, 3(6), pp.401-410.
- Hahnfeldt, P., Panigrahy, D., Folkman, J. and Hlatky, L., 1999. Tumor development under angiogenic signaling: a dynamical theory of tumor growth, treatment response, and postvascular dormancy. Cancer Research, 59(19), pp.4770-4775.
- Naumov, G.N., Folkman, J., Straume, O. and Akslen, L.A., 2008. Tumor‐vascular interactions and tumor dormancy. Apmis, 116(7‐8), pp.569-585.
- Pérez-García, V.M., Calvo, G.F., Bosque, J.J., León-Triana, O., Jiménez, J., Pérez-Beteta, J., Belmonte-Beitia, J., Valiente, M., Zhu, L., García-Gómez, P. and Sánchez-Gómez, P., 2020. Universal scaling laws rule explosive growth in human cancers. Nature Physics, 16(12), pp.1232-1237.
- Superlinear growth reveals the Allee effect in tumors, Youness Azimzade, Abbas Ali Saberi, and Robert A. Gatenby Phys. Rev. E (accepted).
- I. Bozic, C. Paterson, and B. Waclaw, PLoS Computational Biology 15, e1007368 (2019).
© 2026 - The Mathematical Oncology Blog