A spatial game to simulate evolutionary treatments
An evolutionary game theory approach to the emergence of treatment-resistance
As an Applied Math student at Dartmouth College, I’ve always been interested in ways that math can be a powerful tool for solving important real-world problems, so when I came across the opportunity to spend a semester in Tampa doing mathematical oncology research at Moffitt, I was intrigued and excited. Though I hadn’t taken a biology class in a while and didn’t know much cancer biology, I applied because I was interested in seeing firsthand how the mathematical and computational tools I had been taught were actually being employed outside of the classroom. As my first-time doing research, it was exciting to see the way that mathematical techniques could be on the forefront of important medical discovery. It was cool to see firsthand the way that mathematical modelling in tandem with experimental research has become imperative to understanding the many complexities of cancer. As someone hoping to do something meaningful with my passion for math, it was inspiring to see the clinical impact of mathematical oncology and its potential to generate important biological insights about cancer evolution. In cancer, intratumor heterogeneity is crucial to tumor evolution, as the more diverse a tumor is, the better it’s able to adapt to different situations. Cancer cells consist of many different genotypes and phenotypes that can lead each other to be quite different from one another and understanding this intra-tumor diversity and the different traits that emerge from this diversity is key to understanding how tumors evolve1. In the context of drug resistance, it’s been seen that the majority of a tumor before a drug is first applied is phenotypically vulnerable to the drug, but there exists a small population of tolerant cells, also called persistors, who are not fully resistant to the drug, but merely able to survive the treatment. These tolerant cells are seen as a precursor to the existence of full on treatment-resistance in a tumor. Additionally, it’s been seen experimentally that stromal cells can dramatically reduce the ability of a drug to kill tumor cells that might otherwise be vulnerable to the treatment. Working on my own project, I created a spatial evolutionary game to better understand the emergence of treatment resistance from an evolutionary game theory perspective. The emergence of treatment resistance is a key problem in cancer treatment, as initially effective treatments often fail in the long term as tumors eventually develop some resistance to said treatment. Evolutionary Game Theory, a mathematical framework for understanding Darwinian competition, is an appropriate tool for investigating this phenomenon, as the emergence of treatment resistance can be seen as a product of natural selection. Evolutionary Game Theory models are particularly appropriate when the aim is to model cell to cell interactions between various phenotypes and frequency-dependent selection. My project assumes insights that are based on emerging experimental discoveries of stromal protection and the existence of treatment tolerant (persistor) cells as a precursor to full-on treatment-resistance, and these insights are encapsulated in an evolutionary game that allows us to better understand the dynamics of treatment-resistance in various contexts. The evolutionary game I created models the interplay between three different types of tumor cells: treatment-vulnerable, treatment-tolerant (persistors), and treatment-resistant cells. We include stromal cells in the model but not as players in the game to incorporate the effect of stromal protection of tumor cells that might otherwise be affected by treatment. Our game is encapsulated in the payoff table below:
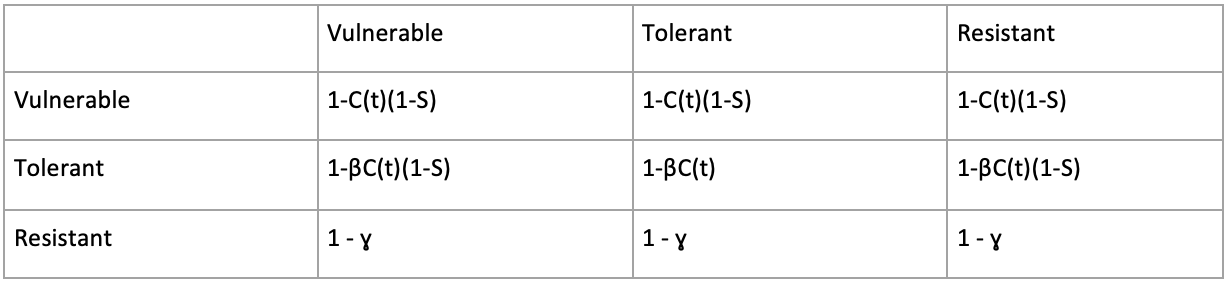 Treatment-resistant cells are considered to be completely unaffected by treatment, but they pay a fitness cost for having resistance, thus the evolutionary fitness they get from every interaction is 1 - ɣ, where ɣ is the cost of being treatment-resistant. For both vulnerable and tolerant cells that don’t pay a cost of resistance, both get a payoff of 1 if no treatment is being applied. Otherwise, if treatment is on, these cells pay a fitness cost from treatment, represented by C(t). If tolerant or vulnerable cell populations are near the stroma when treatment is being applied, they pay less of a fitness cost during treatment, as the variable S represents the extent to which stromal protection is received, or how close the cell is to the surrounding stroma. Tolerant cells, who are less vulnerable to treatment than the vulnerable cells, take less of a fitness cost from treatment, as β (in between 0 and 1) represents how much less affected tolerant cells are by treatment.
Using HAL, I created a spatial implementation of our game in the form of an agent based model, allowing us to simulate the effects of evolutionary treatment strategies.
We created a 2D grid for cells of each population to play the game on, initializing the grid with a tumor of treatment-tolerant and treatment-vulnerable cells and surrounding stroma (see figure below). At each timestep, each cell plays the game with its neighbors, and if it has room to reproduce, the cell’s chance of successfully reproducing is proportional to its average fitness from the game. Additionally, every time a tolerant cell reproduces, it has a small chance of mutating into a resistant cell, so the resistant population emerges randomly as a mutation of the tolerant cell population. Treatment has the effect of raising the death rates of vulnerable and tolerant cells, as well as changing the game above.
The gif shows an example of tumor growth from our game, where treatment is applied continuously. The grid is grey when treatment is being applied and white represents the surrounding stroma. After treatment starts (screen goes gray), the vulnerable cells (green) which are initially the majority of the tumor population die out, and the tolerant cells (blue) start to emerge as the majority of the tumor until a mutation occurs and the resistant population (red) takes over.
Treatment-resistant cells are considered to be completely unaffected by treatment, but they pay a fitness cost for having resistance, thus the evolutionary fitness they get from every interaction is 1 - ɣ, where ɣ is the cost of being treatment-resistant. For both vulnerable and tolerant cells that don’t pay a cost of resistance, both get a payoff of 1 if no treatment is being applied. Otherwise, if treatment is on, these cells pay a fitness cost from treatment, represented by C(t). If tolerant or vulnerable cell populations are near the stroma when treatment is being applied, they pay less of a fitness cost during treatment, as the variable S represents the extent to which stromal protection is received, or how close the cell is to the surrounding stroma. Tolerant cells, who are less vulnerable to treatment than the vulnerable cells, take less of a fitness cost from treatment, as β (in between 0 and 1) represents how much less affected tolerant cells are by treatment.
Using HAL, I created a spatial implementation of our game in the form of an agent based model, allowing us to simulate the effects of evolutionary treatment strategies.
We created a 2D grid for cells of each population to play the game on, initializing the grid with a tumor of treatment-tolerant and treatment-vulnerable cells and surrounding stroma (see figure below). At each timestep, each cell plays the game with its neighbors, and if it has room to reproduce, the cell’s chance of successfully reproducing is proportional to its average fitness from the game. Additionally, every time a tolerant cell reproduces, it has a small chance of mutating into a resistant cell, so the resistant population emerges randomly as a mutation of the tolerant cell population. Treatment has the effect of raising the death rates of vulnerable and tolerant cells, as well as changing the game above.
The gif shows an example of tumor growth from our game, where treatment is applied continuously. The grid is grey when treatment is being applied and white represents the surrounding stroma. After treatment starts (screen goes gray), the vulnerable cells (green) which are initially the majority of the tumor population die out, and the tolerant cells (blue) start to emerge as the majority of the tumor until a mutation occurs and the resistant population (red) takes over.

Continuous Application of Treatment
In this model, we slowly see the emergence of treatment resistance due to natural selection, and from this we are able to simulate the effects of evolutionary treatment strategies. The goal of evolutionary treatment strategies is to provide an alternative to the conventional administration of a maximal tolerable dose of a drug, which quickly leads to the emergence of treatment-resistance. The hope is that by treating tumors in a way that accounts for tumor evolution, we can slow down or prevent the emergence of treatment resistance to improve long-term patient outcomes. Adaptive therapy is an evolutionary treatment strategy that attempts to select against the emergence of treatment resistance where the goal is not to completely eradicate the tumor, but to apply treatment intermittently in a way that resistant cells never emerge as the dominant phenotype, so that you can indefinitely control the size of the tumor so that it never becomes lethal. I implemented a form of adaptive therapy2 in which I try to control the size of the tumor within a specific size-range, where I start applying treatment if the size of the tumor reaches the upper bound of our target size-range, and I stop applying treatment when the size of the tumor decreases below the lower bound of our target size-range. Eventually, the population of fully resistant cells emerges in the tumor, so we aren’t able to control the size of our tumor between our desired range forever. Nonetheless we found that by having treatment breaks that give tolerant (blue) and vulnerable cells (green) a temporary advantage over the resistant cell population (red), this strategy of adaptive therapy does better than the current standard of treatment, administering treatment in a continuous fashion. Below is an example of our form of adaptive therapy (treatment is being applied when the grid is grey):
Adaptive Therapy
A significant result found in our spatial game was that when we change the cost of treatment in our game, corresponding to a lower or higher dose of treatment, we saw that when applying adaptive therapy, smaller doses work better during early stage tumor growth, while larger doses work better during later stages. The figures below compare how long it takes on average for the model to grow a tumor to a size of 70,000 cells with continuous application of treatment (blue bars) vs adaptive therapy (red bars). The left figure shows the results with a lower dose, or a lower cost of treatment, and the right shows our results with a higher dose, and you can see that adaptive therapies with a higher dose do better when treatment is applied at a later stage, while with a lower dose, adaptive therapies do much better earlier.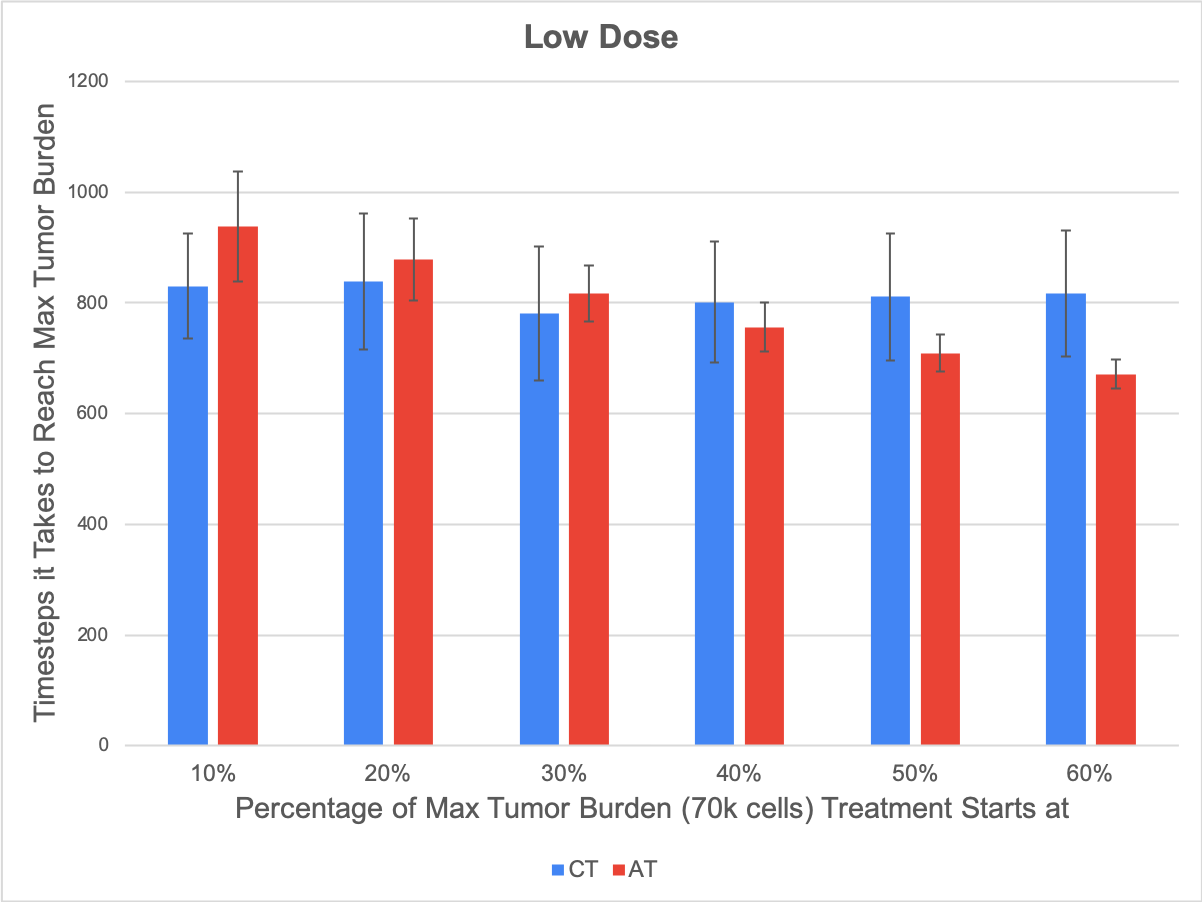
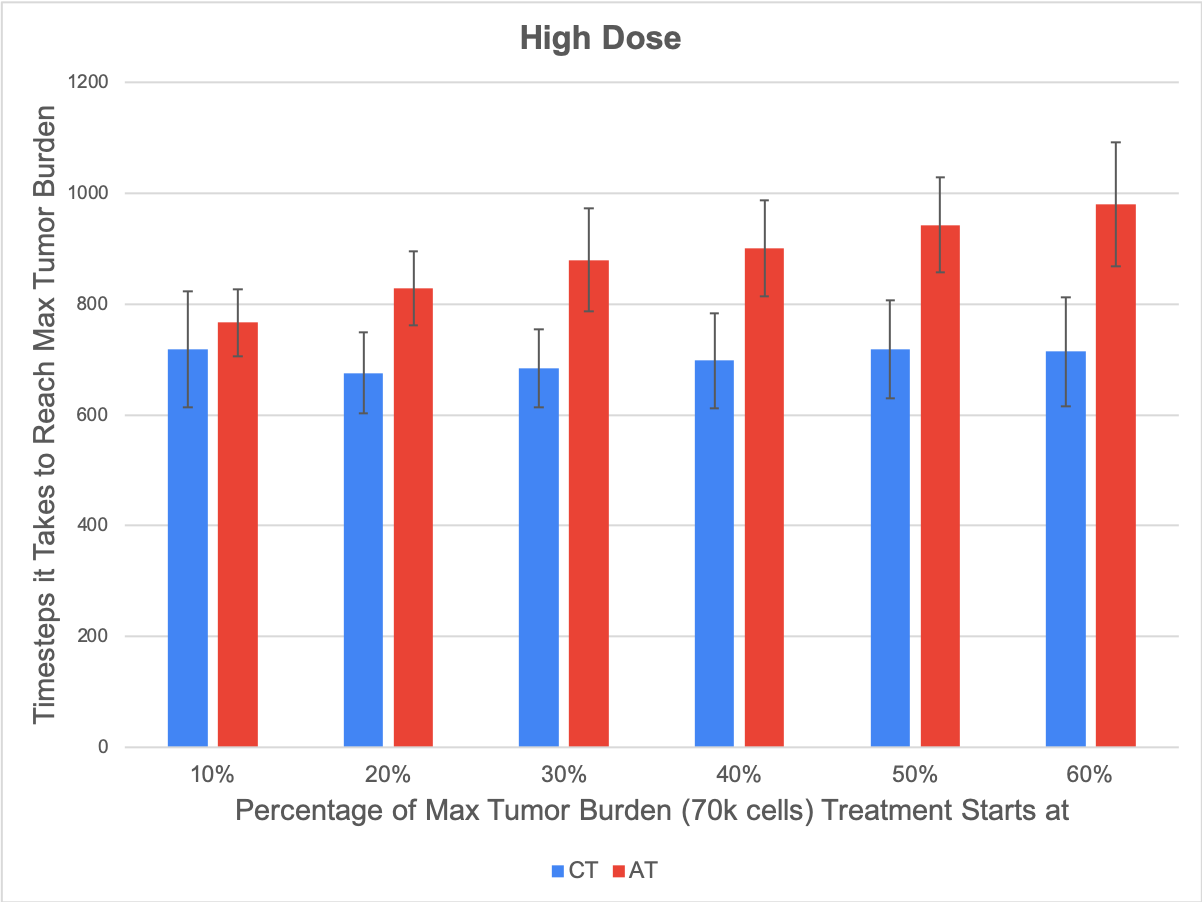
Figure 1: Adaptive therapy vs continuous treatment strategies with a high dose (right) and a low dose (left). Each bar corresponds to the average number of timesteps it took for our tumor to grow in 50 simulations to our max burden (a size of 70,000 cells). The blue bars show the results for continuous application of treatment, while the red bars show the results for our mode of adaptive therapy. The x-axis values correspond to the size of the tumor (as a percentage of our max burden) at which we start applying treatment.
Another thing we investigated was the effect of stromal protection on the emergence of treatment-resistance, so we looked at what happened in our model when we varied the concentration of stroma in our grid. We saw that the only effect that increasing the amount of stroma had was that it limited the space the tumor had to grow. Below are some results from our low stroma case (left) compared with our high stroma case (right) with a high dose of treatment. We saw that regardless of the concentration of stroma, the same general pattern emerges, where applying adaptive therapy does better than continuous treatment, and the margin by which adaptive therapy does better increases the later that you start treatment. In our model, the stroma didn’t have any effect on which treatment strategies were optimal, and the only relevant effect of stromal protection was that it limited the space our tumor had to grow.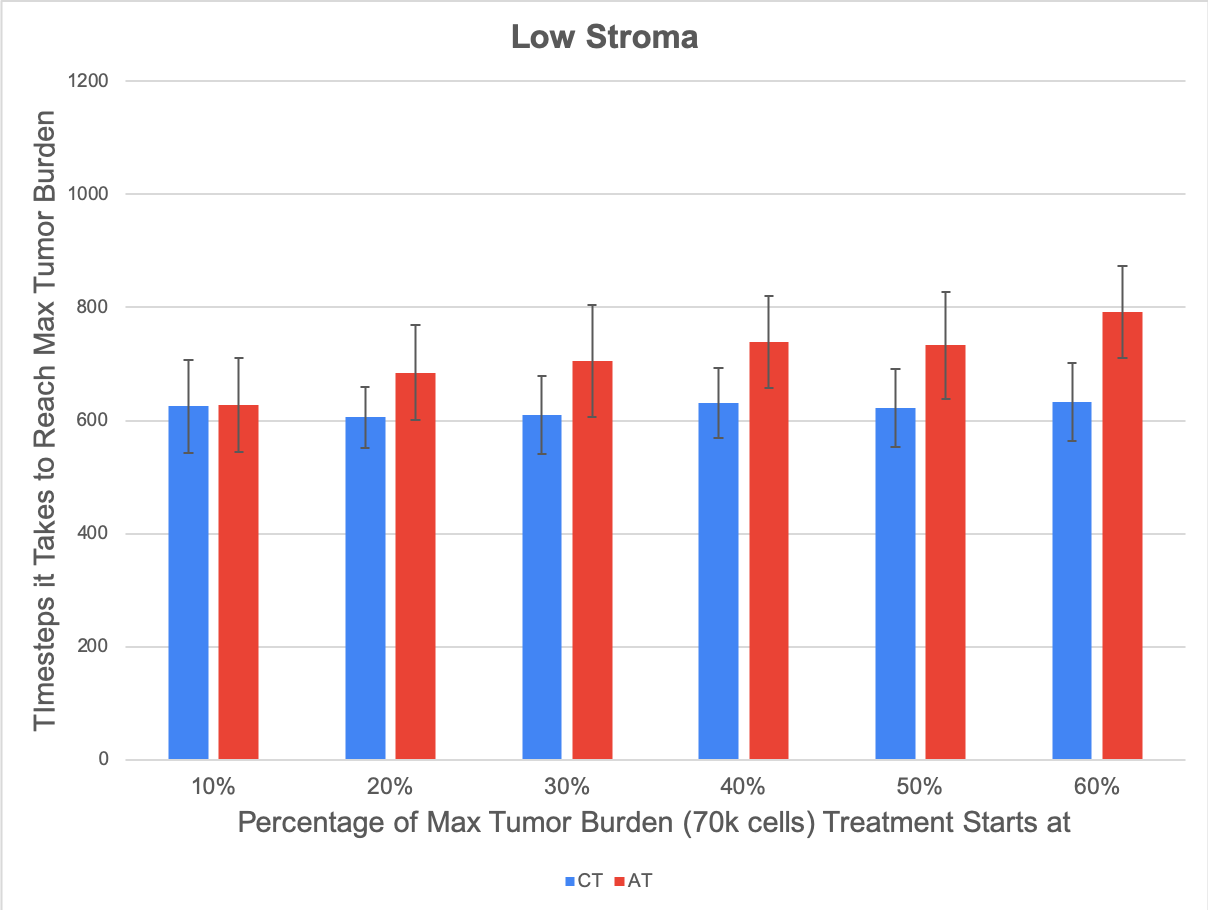
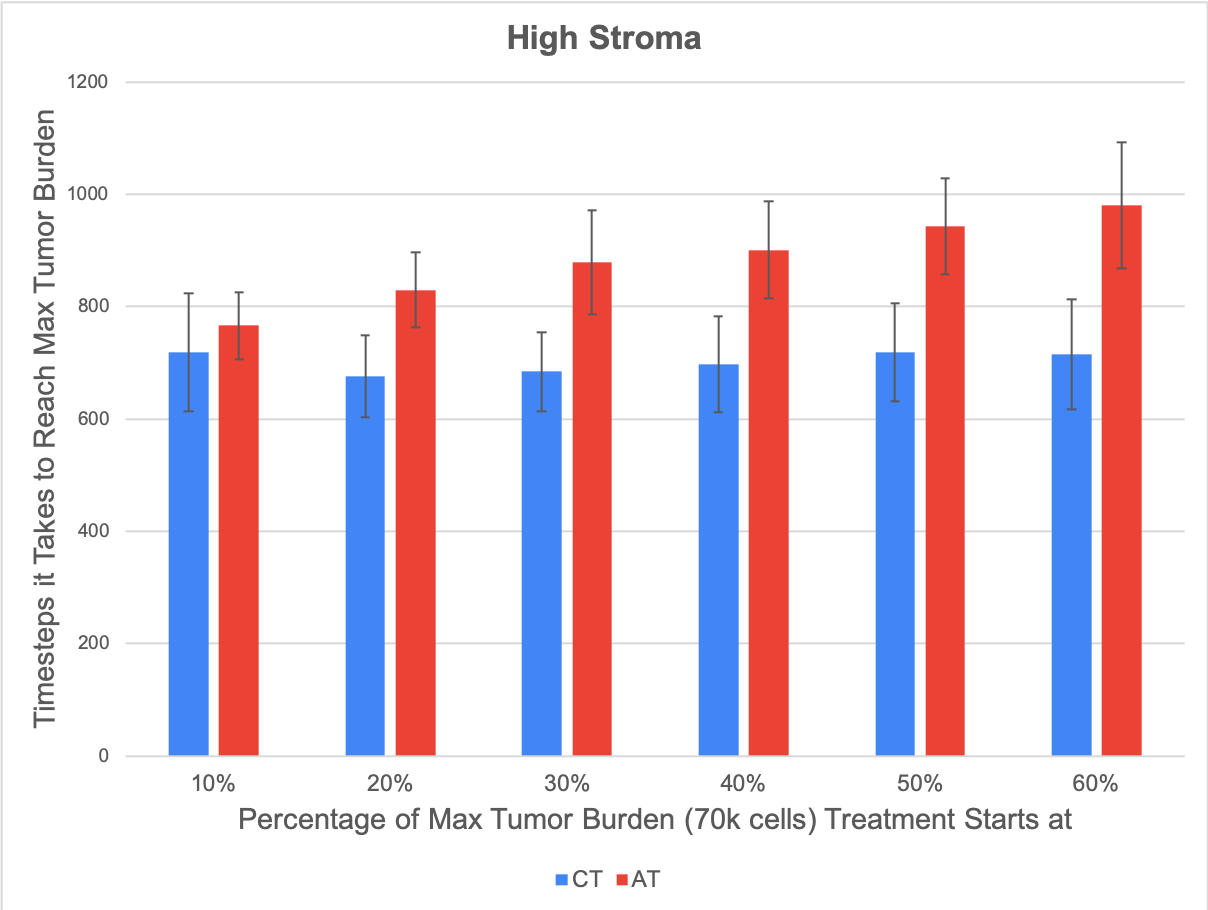
Figure 2: Low vs High concentration of stroma. Stromal protection had no effect on the dynamics of treatment resistance, as the only significant effect of introducing more stroma into the game was that it slowed down overall tumor growth by limiting the open space the tumor had to grow, but there was no change in which treatment strategies were optimal.
The model initially was created to study the emergence of treatment resistance in TK-inhibitors, so we made the assumption that treatment targets all cells equally, regardless of whether they were actively proliferating or not, but for other types of treatments such as cytotoxic chemotherapy, it may be more realistic to consider that treatment only affects the cells that are actively dividing. A final significant result we found from our game was that adaptive therapy may be more suitable for types of treatments that target actively proliferating cells on the growing edge of a tumor. The difference in the results of changing how treatment is applied are shown in the figure below. The plots compare the average time it takes for our model to grow a tumor of 70,000 cells with continuous treatment and adaptive therapy strategies. When we changed our model to make treatment only affect the outside core of the tumor (cells that have open slots near them and can proliferate), we found that the margin by which adaptive therapy (shown in red) did better than continuous treatment (shown in blue) was much greater than when treatment was applied in a way that affected all tumor cells equally.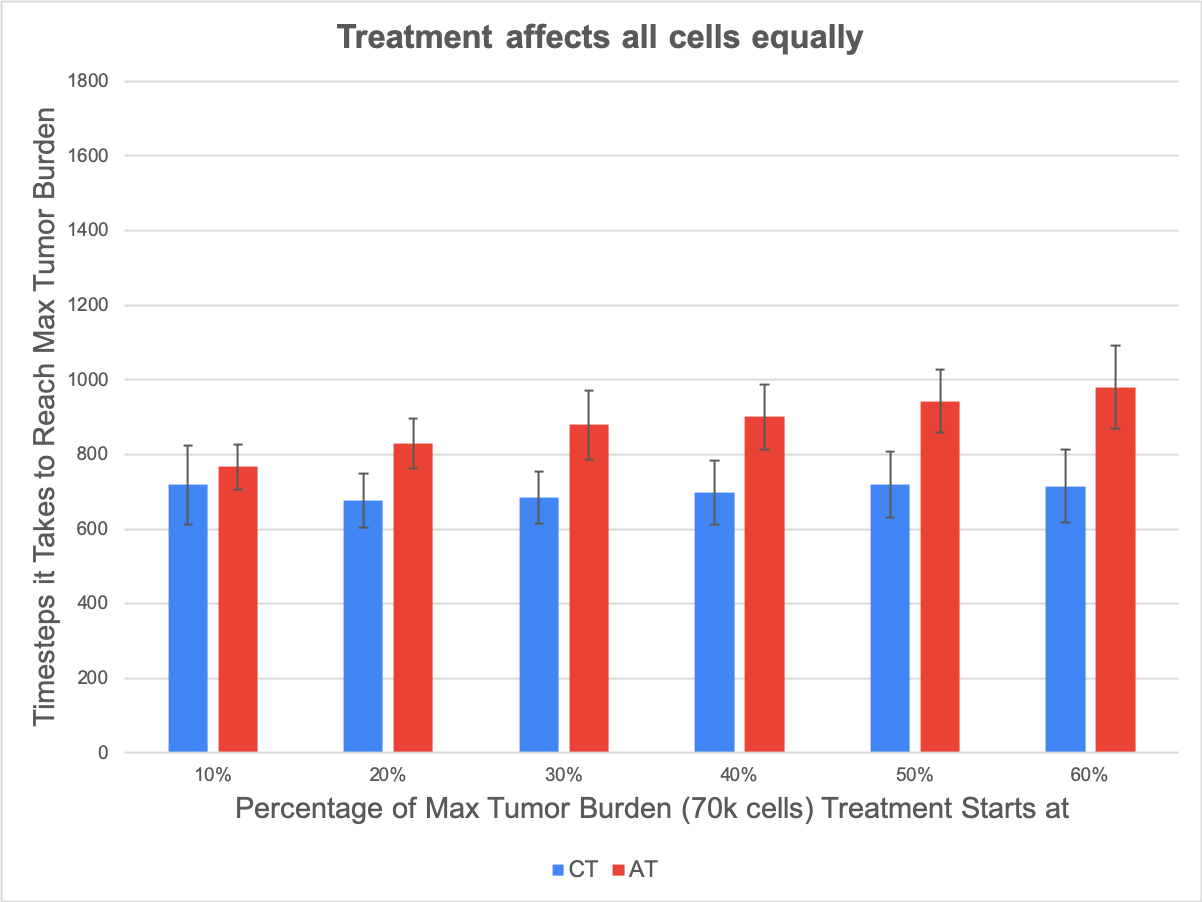
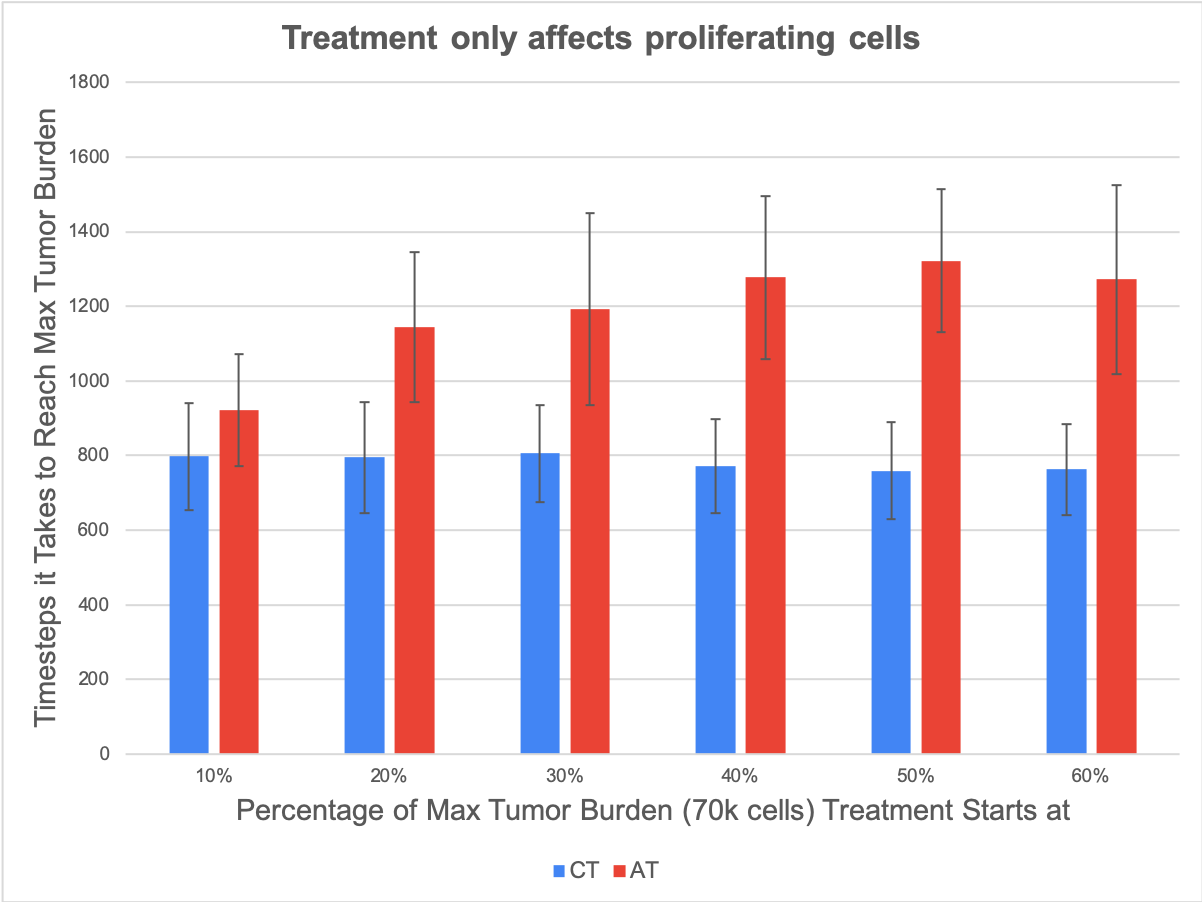
Figure 3: Changing the way that treatment is applied. Adaptive therapy is much more successful in comparison to continuous treatment when treatment is applied in a way that only affects proliferating cells, as opposed to when treatment is applied in a way that affects all cells equally.
Overall, by creating a spatial game between tumor subpopulations of different levels of treatment resistance, I was able to simulate the emergence of treatment-resistance under different contexts, allowing me to test evolutionary therapies. The results indicate that adaptive therapies could be more successful at limiting overall tumor growth than continuous treatment application by slowing down the emergence of treatment resistance, and saw that for treatments that only affect the outside core of a tumor (e.g. cytotoxic chemotherapy), adaptive therapy may be more suitable (see figure 3) than it is for treatments that affect all cells in a tumor equally (e.g. TK-inhibitors). Moreover, we found that adaptive therapy is more successful with a lower dose at an earlier stage of tumor growth, while adaptive therapy is more successful with a higher dose at a later stage in tumor growth (see figure 1). One final and interesting result, considering that I was aiming to explore the role of stromal protection is that, other than slowing down growth by limiting the space that a tumor has to proliferate, the stroma did not seem to provide the impact I expected on these results. This could suggest a new dimension to the role of carcinoma associated fibroblasts as they do protect the tumor against treatment but also compete for space. Importantly, it could also point to a limitation of the 2D nature of the agent-based model and the lattice-based approach of the implementation. In reality, 3D allows for more opportunities for stroma and tumor to coexist and we expect that some of the results I presented here would be attenuated in an in vivo context. Within my results, it’s worth noting that there’s never a scenario in which the tumor is completely eradicated, which can be seen as a product of the assumption that treatment has no effect on the phenotypically resistant cell population, a population that will inevitably arise. Fundamental to this model is the notion that once a certain level of treatment-resistance emerges, the tumor will grow even when treatment is being applied. Thus, the fact that treatment-resistance is eventually inevitable makes these results more suitable for understanding later stage tumors that may not be entirely curable, and this is why the metric used to measure the success of different strategies is the time it takes for the tumor to grow to a substantial size of 70,000 cells. While this study is more geared towards situations in which treatment-resistance is eventually inevitable, the framework for this spatial game is generalizable to different contexts, and we expect that by modifying some of the assumptions about resistant cells, for example, or introducing two types of treatments into the game to explore the concept of the evolutionary double bind, there’s room for further exploration and the potential to explore different situations. My winter spent at IMO taught me a lot about the promising mathematical and computational approaches being used to understand cancer. Many thanks to Dartmouth’s Dorothy Wallace and Moffitt’s Heiko Enderling for putting the program together, as well as to David Basanta for the supervision and guidance with my project. As an undergraduate interested in all of the research being done that proves that math is important, working at IMO was an incredible and fascinating experience, and it was amazing to see all of the cool and important things math can do when it comes to cancer.References
- Gerlinger, Marco et al. “Intratumor Heterogeneity and Branched Evolution Revealed by Multiregion Sequencing.” New England Journal of Medicine, vol. 367, no. 10, 2012, pp. 976–976., doi:10.1056/nejmx120044.
- Gatenby, Robert A., et al. “Adaptive Therapy.” Cancer Research, American Association for Cancer Research, 1 June 2009, cancerres.aacrjournals.org/content/69/11/4894.
© 2026 - The Mathematical Oncology Blog